Key words: dyslipidemia, insulin resistance, coronary artery disease, prostate cancer survivors, statin-intolerance, ursodeoxycholic acid
Ключові слова: дисліпідемія, інсулінорезистентність, ішемічна хвороба серця, пацієнти, що вижили після раку передміхурової залози, непереносимість статинів, урсодезоксихолева кислота
Abstract
Numerous studies suggest an increased frequency of dyslipidemia and metabolic disorders in patients with prostate adenocarcinoma. The aim was to evaluate lipid profile, insulin resistance level in men with coronary artery disease and concomitant prostate cancer and impact of ursodeoxycholic acid on these indicators in complex therapy. Study included 48 men with coronary artery disease and prostate adenocarcinoma. The lipid profile, insulin resistance, testosterone level were determined in all of patients. The level of insulin, C-reactive protein were determined by immune-enzyme analysis. 1st group (n=22) was made up by statin-intolerant patients treated with ursodeoxycholic acid during 60 days in addition to standard therapy; 2nd group (n=26) – by statin-intolerant patients treated with standard regimen. The levels of triglycerides, low density lipoprotein-cholesterol and apolipoprotein-B in men with coronary artery disease and prostate adenocarcinoma were significantly higher than those in control group (p<0.001). The complex therapy with ursodeoxycholic acid resulted in significant decrease in the level of total cholesterol by 17.6% (p<0.001), triglycerides by 21.6% (p<0.001), low density lipoprotein-cholesterol by 25.2% (p<0.001), apolipoprotein-B by 12.9% (p<0.001), HOMA index level by 19.4% (p=0.001), median insulin level by 22.9% (p=0.002). Men with coronary artery disease and concomitant prostate cancer are characterized by increased levels of triglycerides, low density lipoprotein-cholesterol, insulin resistance and decreased apolipoprotein-B associated with low testosterone level. Complex treatment with ursodeoxycholic acid in these patients contributed to significant lipid level improvement and additional decrease of insulin resistance.
Реферат
Метаболічні зміни в чоловіків з ішемічною хворобою серця і супутнім раком передміхурової залози та можливості їх корекції в пацієнтів з непереносимістю статинів. Курята О.В., Сіренко О.Ю. Численні дослідження свідчать про підвищену частоту дисліпідемії та метаболічних порушень у пацієнтів з аденокарциномою простати. Метою дослідження було оцінити ліпідний профіль, рівень інсулінорезистентності в чоловіків з ішемічною хворобою серця та аденокарциномою простати та вплив урсодезоксихолевої кислоти на ці показники в комплексній терапії. У дослідженні взяли участь 48 чоловіків з ішемічною хворобою серця та аденокарциномою простати. У всіх пацієнтів визначали ліпідний профіль, інсулінорезистентність, рівень тестостерону. Рівень інсуліну, С-реактивного білка визначався за допомогою імуноферментного аналізу. 1-шу групу (n=22) склали пацієнти з непереносимістю статинів, які отримували урсодезоксихолеву кислоту протягом 60 днів як доповнення до стандартної терапії; 2-гу групу (n=26) – пацієнти з непереносимістю статинів, які отримували стандартний режим лікування. Рівні тригліцеридів, холестерину ліпопротеїнів низької щільності та аполіпопротеїну-B у чоловіків з ішемічною хворобою серця та аденокарциномою передміхурової залози були достовірно вищими, ніж у контрольній групі (p<0,001). Комплексна терапія із включенням урсодезоксихолевої кислоти привела до достовірного зниження рівня загального холестерину на 17,6% (p<0,001), тригліцеридів на 21,6% (p<0,001), холестерину ліпопротеїнів низької щільності на 25,2% (p<0,001), аполіпопротеїну-B на 12,9% (p<0,001), рівня індексу HOMA на 19,4% (p=0,001), інсуліну на 22,9% (p=0,002). Чоловіки з ішемічною хворобою серця та аденокарциномою простати характеризуються підвищеним рівнем тригліцеридів, холестерину ліпопротеїнів низької щільності, інсулінорезистентністю та дефіцитом аполіпопротеїну-B, що асоціюється з низьким рівнем тестостерону. Комплексне лікування із включенням урсодезоксихолевої кислоти в цих пацієнтів сприяло достовірному поліпшенню показників ліпідного профілю та додатковому зменшенню рівня інсулінорезистентності.
Cardiovascular diseases (CVD) and oncological diseases continue to be the main causes of mortality worldwide. Improvements in cancer survival have led to greater number of successfully treated patients with an increased mortality from cardiovascular diseases and their complications [1].
Prostate cancer (PC) is associated with increasing incidence rates worldwide and it is the second most common cancer in men [3]. Coronary Artery Disease (CAD) is prevalent and one of the leading cause of mortality in men with prostate cancer [2]. Increasing evidence of overlapping risk factors for cancer and cardiovascular disease in patients with PC represents a relevance of the cardio-oncological subspecialty for optimization of survivorship outcomes [4].
Numerous studies suggest an increased frequency of cardiovascular diseases and metabolic disorders in patients with prostate adenocarcinoma [5, 6], therefore, correction of risk factors is relevant, especially of dyslipidemia and insulin resistance (IR) [7, 11]. As known, the main pharmacological agents for treatment of dyslipidemia are statins [8, 21]. However, treatment with statins not always leads to achievement of lipid level targets, especially triglycerides [12]. According to literary data, up to 29% of patients report adverse musculoskeletal effects that limit using statins or their guideline-recommended doses [25]. Limitations of using statins for patients with cancer demonstrate conflicting results, increased muscle loss in some models, increased risk of muscle pain in elderly patients and drug interactions [22]. Therefore, in recent years, there has been active interest in other groups of lipid-lowering drugs. Thus, latest trials have shown bempedoic acid efficacy in lowering LDL cholesterol and its ability to reduce cardiovascular events in statin-intolerant patients [23], but has limited data for cancer patients, though ongoing research [26].
In recent literature, increasing attention is being paid to the possibilities of using ursodeoxycholic acid (UDCA) in patients with cancer [9]. It was found, that UDCA can protect epithelial cells from damage and apoptosis, while suppressing the proliferation and induction of apoptosis of cancer cells, including in prostate adenocarcinoma. Therefore, there is evidence that UDCA has a positive effect on the treatment of cancer patients, in particular, with hepatotoxicity caused by chemotherapy [13]. Interestingly, that UDCA has some hypocholesterolemic effect, reduces cholesterol synthesis by decreasing bile secretion and inhibiting the absorption of cholesterol in the intestine [10].
Thus, the purpose of the present study was to evaluate lipid profile, insulin resistance level in men with coronary artery disease after prostate cancer treatment and impact of UDCA on these indicators in complex therapy.
MATERIALS AND METHODS OF RESEARCH
The present study was conducted according to the Helsinki declaration principles and was approved by the Biomedical Ethics Commission of the Dnipro State Medical University (protocol No. 24 dated 01/15/25). All participants of research gave informed written consent. Dynamic observation and treatment was performed in 48 men with CAD with concomitant prostate adenocarcinoma (stages I-IIІ) at the age of 45 to 75 years (the main group). The baseline characteristics of patients have been demonstrated in the Table 1.
All patients had verified CAD according to the guidelines of the European Society of Cardiology, the diagnosis of prostate cancer was confirmed by a biopsy. Аll patients received standard treatment of CAD but stopped taking statins due to side effects or refused to take them. The inclusion criteria were: men aged 45-75 years, verified diagnosis of CAD and prostate cancer (stages I-IIІ), completed course of anticancer therapy by the doctor's decision (due to unacceptable toxicity) or the patient's refusal more than 6 months ago, consistently selected cardiologic treatment for the last 3 months, voluntary informed consent to participate in the study.
The presence of CAD was assessed based on documented cardiovascular disease, such as a history of myocardial infarction, angiographically confirmed coronary artery stenosis, and a history of coronary artery revascularization procedures.
According to magnetic resonance imaging (MRI) data, 40 (83,3%) of patients had tumors localized to the prostate gland without extracapsular extension (T1c-T2b), while 8 (16,7%) had tumors of a higher grade (T3a-T3b).
Exclusion criteria from the study were: age over 75 years; previous therapy with statins within the last 3 months, arrhythmias that cause hemodynamic disturbances and require correction with antiarrhythmic agents (including high gradations of ventricular arrhythmias, atrial flutter, paroxysmal tachycardias, sinus node weakness syndrome, atria-ventricular block 2-3 degrees), acute myocardial infarction, acute cerebrovascular accident, diabetes, hyper- and hypothyroidism with thyroid stimulating hormone (TSH) levels >4 mU/l, chronic kidney insufficiency (glomerular filtration rate (GFR) <30 ml/min/1.73 m2), acute heart failure, obesity with grade 4, cachexia.
Characteristic Men with coronary artery disease after prostate cancer (n=48) Control group (n=31) p Median of age, years 67 [56.8; 72.7] 65.4 [57.4; 74.2] 0.651 Median duration of CAD, years 9.8 [7.6; 10.4] 9.5 [7.5; 9.7] 0.143 Glomerular filtration rate (GFR), ml / (min · 1.73 m2) 76 [63; 79] 78 [63.8; 81.2] 0.177 Patients with stable angina, % 64.6 54.8 0.461 Patients with previous MI, % 29.2 29.0 0.214 Patients with hypertension, % 85.4 80.6 0.614 Median of ejection fraction, % 62.8 [51.4; 68.4] 65.1 [52.6; 69.6] 0.158 Patients, received cardiology treatment (%): ACE inhibitors/ACE receptors blockers 79.2 77.4 0.122 aldosterone antagonists 75.0 67.7 0.092 β-blockers 79.2 80.6 0.266 calcium antagonists 72.9 61.3 0.347 antiplatelet agents 68.8 67.7 0.284 diuretics 54.2 48.4 0.521
Patients were blindly divided into two groups: I group (n=22) – patients with CAD and prostate cancer treated with UDCA (Ursofalk, Dr. Falk Pharma GmbH, Germany) during 60 days in addition to standard therapy. The dose was calculated individually – 15 mg/kg. Group II (n=26) – patients who were treated with standard regimen without agreement on UDCA therapy. The control group consisted of 31 men with CAD without prostate cancer, comparable in age, cardiovascular risk profile, obtained by treatment of CAD.
At the initial state, the patients of both groups were comparable in terms of the main disease, indicators of age, duration of the disease, level of GFR, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), triglycerides (TG), systolic and diastolic blood pressure, heart rate.
All the patients were examined by physician, laboratory and instrumental methods at the beginning and to the end of the study were used. The patients were observed in outpatient department. The standard clinical and biochemical laboratory tests, blood tests for detection of total cholesterol (TC), TG, LDL-C, HDL-C, apolipoprotein А (Аpо-А1), apolipoprotein В (Аpо-В) [28], serum insulin [29] were performed. Tolerability of the drug was determined on the basis of an assessment of laboratory parameters (the level of ALT, AST in the serum), the level of GFR, the incidence of adverse reactions. GFR was determined by the formula CKD-EPI [30], IR – by the formula HOMA [14].
In order to evaluate the state of androgen deficiency the testosterone level was determined by the method of immunoenzymatic analysis with the reagent test kit “AccuBind ELISA”, androgen deficiency were established in case of total testosterone level below 2.5 ng/ml.
Statistical processing of the obtained results was performed using the licensed program STATISTICS 12.0 (StatSoft Inc, USA, trial version). Non-parametric statistics were used [27]. The data was presented in the form of a median (Me) and the interquartile segment [25%; 75%]. Categorical data were presented as n (%) and were compared using Chi-squared test. For comparison of indicators in two independent groups, the Mann-Whitney U-test, the two-sided Fisher exact test, and the Wilcoxon test (W) were used to compare two dependent groups. Statistically significant differences in research results were determined at a level of p<0.05.
RESULTS AND DISCUSSION
The incidence of dyslipidemia was significantly higher in main group compared to control group (p< 0.001). The levels of TG, LDL-C and Apo-B in men with CAD and concomitant prostate cancer were significantly higher than those in control group (Table 2) (p< 0.001). Significant correlation relations between the testosterone level with TG, HDL-C, LDL-C, Apo-B were determined in men with CAD and concomitant prostate cancer – R= -0.58 (p<0.001), R=0.60 (p<0.05), R= -0.74 (p<0.001), R= -0.79 (p<0.001) respectively.
Parameter Men with CAD and prostate adenocarcinoma (n = 48) Men with CAD (n = 31) median [25%;75] (mmol/l) abnormal (n, %) median [25%;75] (mmol/l) abnormal (n, %) TC 6.12 [4.98;6.64] 34(70.80)* 5.78 [4.36;6.15] 16(51.6) TG 3.26 [2.68;3.89]** 38(79.17)* 1.56 [1.18;2.04] 12(38.71) HDL-C 1.43[1.15;1.79] 5(10.42) 1.50[1.10;1.68] 2(6.45) LDL-C 4.53[3.74;4.86]** 39(91.25)* 3.43[3.01;3.95] 20(64.52) Apo-A1 1.4[1.14;1.58] 8(16.67) 1.31[1.20;1.47] 5(16.13) Apo-B 1.88[1.62;2.09]** 25(52.08)* 1.22[1.03;1.54]* 7(22.58) Dyslipidemia, % – 42(87.50) * – 18(58.06) Notes: * – p<0.05; ** – p<0.001 comparison of lipid levels and dyslipidemia incidence between cancer patients and control group; TC: total-cholesterol; TG: triglyceride; HDL-C: high density lipoprotein-cholesterol; LDL-C: low density lipoprotein-cholesterol; Apo-A1: apolipoprotein-A1, Apo-B: apolipoprotein-B.
The level of insulin in patients with CAD and concomitant prostate cancer ranged from 10.7 to 25.9 MO/ml baseline, the median insulin level was 18.9 [11.3; 23.7] MO/ml, in the control group – ranged from 5.4 to 17.5 MO/ml baseline, the median insulin level was 10.8 [5.5; 13.4] MO/ml respectively (p<0.001). IR was established in 29 (60.42%) patients of main group and 13 (41.94%) of controls (p<0.05). Men with CAD and concomitant prostate cancer had significantly higher level of insulin, HOMA index compared with the control group (p<0.001). Correlation relations between the HOMA index with PSA, testosterone level, TG, LDL-C were determined in men with CAD after prostate adenocarcinoma treatment – R=0.65 (p<0.001), R= -0.68 (p<0.001), R=0.58 (p<0.05), R=0.63 (p<0.05) respectively.
The complex therapy with UDCA for patients with CAD and co-existent prostate adenocarcinoma resulted in a more significant reduction of the level of the pro-atherogenic fractions of blood lipids (р<0.01) (Table 3). Thus prescribing only standard therapy contributed to a significant decrease in the level of TC by 12.2% (p<0.001), TG by 18% (p<0.001), LDL-C by 20% (p<0.001), number of dyslipidaemia pts by 23.08% (p<0.001). On the other hand the complex therapy with UDCA resulted in a greater reduction of pro-atherogenic fractions of blood lipids: significant decrease in the level of TC by 17.6% (p<0.001), TG by 21.6% (p<0.001), LDL-C by 25.2% (p<0.001), number of dyslipidaemia pts by 31.82% (p<0.001) and additional significant decrease in the level of Apo B by 12.9% (p<0.001). The significant differences between Group I and II after observation were identified by LDL-C level (p<0.001). There were no significant differences between the levels of liver transaminases before and after observation period in all groups of pts (Table 3).
Lipid profile indicators Before treatment After treatment men with coronary artery disease after prostate cancer treatment (n=22) men with coronary artery disease after prostate cancer treatment -UDCA (n=26) men with coronary artery disease after prostate cancer treatment +UDCA (n=22) men with coronary artery disease after prostate cancer treatment -UDCA (n=26) TC, mmol/l 6.18 [4.88;6.52] 5.98 [4.76;6.45] 5.09 [4.18; 5.32]* 5.25 [4.18;5.54]* HDL-C, mmol/l 1.40[1.11;1.83] 1.46 [1.18;1.96] 1.48[1.15;1.89] 1.52 [1.25;2.10] LDL-C, mmol/l 4.56[3.74;4.88] 4.50[3.10;4.68] 3.41[3.14;3.65]* 3.60[2.60;4.15]** TG, mmol/l 3.24 [2.65;3.88] 3.33[3.01;3.95] 2.54 [1.48;3.09]* 2.73[2.31;3.34]* Apo-A1 1.42[1.16;1.59] 1.31[1.20;1.47] 1.42[1.15;1.60] 1.35[1.21;1.49] Apo-B 1.86[1.60;2.03] 1.79[1.53;2.14] 1.62[1.51;1.96]* 1.72[1.44;2.00] Dyslipidemia, % 22(100) 26(100) 15(68.18) * 20(76.92)* ALT, U/l 21 [14.78; 28.78] 19 [12.32; 26.75] 24.63 [15.36; 29.79] 22.61 [14.33; 27.77] AST, U/l 26.14 [16.29; 31.66] 24.54 [13.32; 29.71] 27.52 [17.31; 30.63] 25.51 [14.35; 28.69] Notes: * – p<0.05; ** – p<0.001 comparison of the status of lipidemia between groups; TC: total-cholesterol; TG: triglyceride; HDL-C: high density lipoprotein-cholesterol; LDL-C: low density lipoprotein-cholesterol; Apo-A1: apolipoprotein-A1; Apo-B: apolipoprotein-B; UDCA: ursodeoxycholic acid.
+UDCA
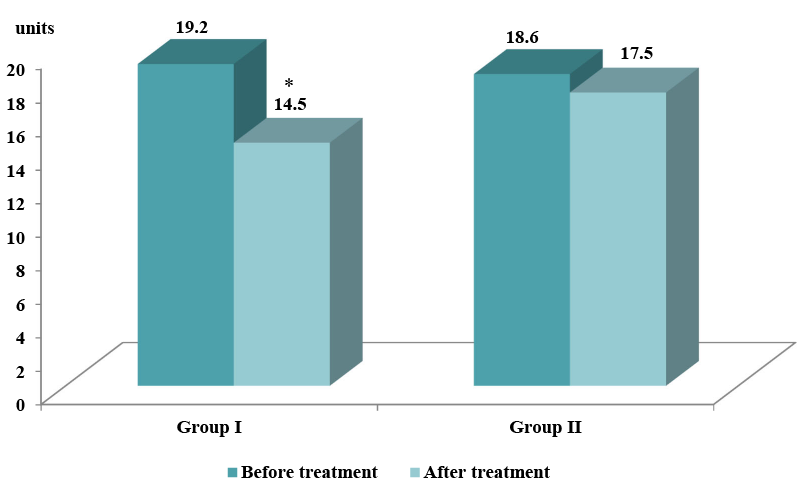
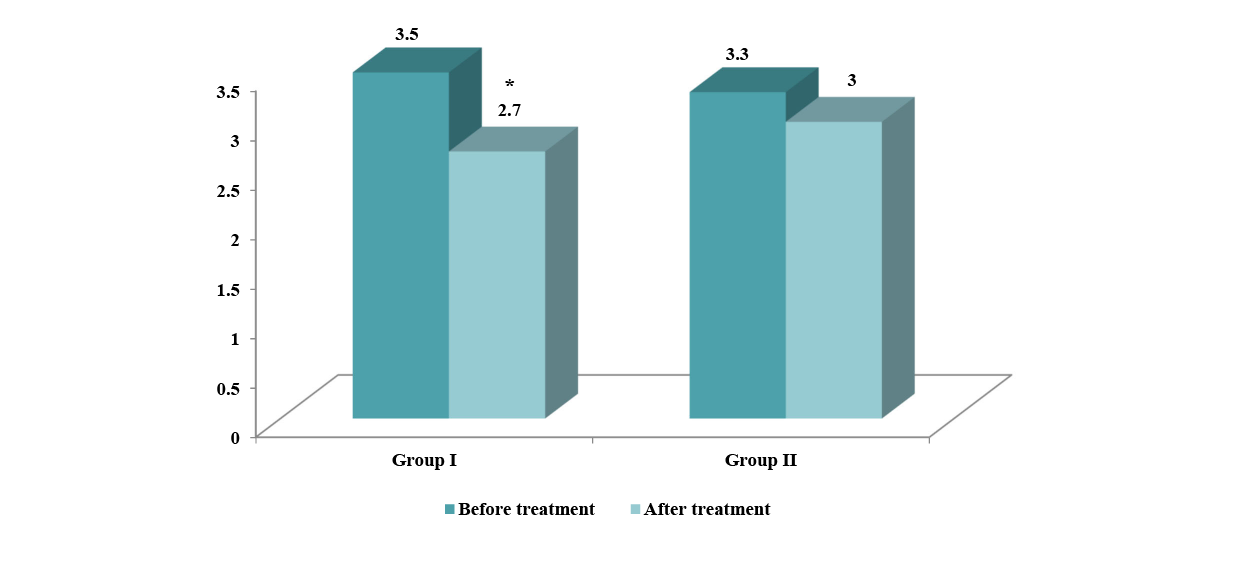
Analysis of the dynamics of the insulin level and insulin resistance on the background of treatment revealed significant differences in these indicators in patients of Group I and II (Fig. 1, 2). In the group of patients UDCA was included in the composition of complex therapy, the median insulin level and HOMA index decreased by 19.4% (p=0.001) and 22.9% (p=0.002), respectively, in the standard treatment group dynamics of these indicator were not significantly different. The inclusion of UDCA in the complex therapy contributed to a more pronounced decrease in insulin level and IR in men with androgen deficiency (Fig. 1, 2). There were no significant side effects with the inclusion of UDCA in the standard therapy in the dynamics of observation, and there was no need to change the daily dose or discontinue treatment. Thus, the results of the study indicate the effectiveness and safety of UDCA in complex treatment in men with CAD with concomitant prostate cancer.
Cancer and CVD are linked by multiple pathways and common risk factors. Thus, their combined treatment is a complex task and requires a multidisciplinary approach with the involvement both of cardiologists and oncologists [15].
Evidence for an association between TC, LDL-C, HDL-C, triglycerides with prostate cancer is conflicting. The retrospective cohort analysis of 843 patients with radical prostatectomy demonstrated association between elevated serum triglycerides with increased risk of prostate cancer recurrence [16]. Cholesterol of LDL-C, HDL-C were not associated with prostate cancer recurrence risk among all of analyzed population. At the same time, among men with prostate cancer and dyslipidemia, elevated total cholesterol and HDL-C levels were associated with increased and decreased risk of cancer recurrence, respectively [16]. Our results demonstrate that the presence of concomitant prostate adenocarcinoma in men with CAD was associated with significantly higher levels of TG, LDL-C and lower Apo-B and were correlated with androgen deficiency.
Thus in recent years a large number of studies suggest the role of lipid and glucose metabolism in the development of prostate cancer [17]. A recent meta-analysis showed a strong positive association between obesity and the risk of advanced prostate cancer. However, epidemiological data on the relationship between lifestyle risk factors (dyslipidemia, diabetes and IR) and the development and progression of prostate cancer remains inconclusive [17].
We provided evidence supporting a relationship of dyslipidemia, IR, and androgen deficiency in men with prostate cancer. Our findings showed a negative correlation between serum testosterone and TG, LDL‑C, serum insulin, and IR. Conversely, we found that HDL‑C, Apo-B was directly correlated with serum testosterone. It was concluded that low serum testosterone is associated with dyslipidemia and IR in men with CAD and prostate cancer. This could be explained by the sex hormone pathway in obesity [18].
Given that standard pharmacologic treatment may not be sufficient to achieve the recommended lipid target, an alternative treatment may be needed. The recent meta-analysis of randomized placebo-controlled trials demonstrated that ursodeoxycholic acid treatment might be an effective lipid-lowering agent with significant reduction in total cholesterol levels [19]. Thus, it was suggested that ursodeoxycholic acid can reduce cholesterol biosynthesis by reducing the activity of hydroxymethylglutaryl-coenzyme A-reductase and decreasing the intestinal absorption of cholesterol [19]. Ursodeoxycholic acid can also affect adipose tissue by lowering triglyceride levels and increasing esterification and desaturation of fatty acids [20].
Our findings suggest that UDCA in complex treatment men with CAD and concomitant prostate cancer decreases pro-atherogenic lipids and IR level. Further investigation is required to elucidate if observed lipid and IR lowering effects of ursodeoxycholic acid in this patients can contribute to the prevention of cardiovascular events.
Limitations.
However, this study should be interpreted with several limitations. Therefore, only patients with CAD with prostate cancer were chosen for this study, and consequently the results can only be applied to this population. It could be perspectival to evaluate the hard end point with prolongation of this therapy.
CONCLUSIONS
1. The present study have demonstrated that men with coronary artery disease and concomitant prostate cancer are characterized by increased levels of triglycerides, low-density lipoprotein cholesterol, IR and decreased apolipoprotein В associated with low testosterone level.
2. Complex treatment with ursodeoxycholic acid in these patients contributed to significant lipid level improvement and additional decrease of insulin resistance.
Contributors:
Kuryata O.V. – conceptualization, methodology, writing – review & editing, project administration;
Sіrenko O.Yu. – methodology, formal analysis, resources, data curation, writing – original draft, investigation.
Funding. This research received no external funding.
Conflict of interests. The authors declare no conflict of interest.
REFERENCES
1. Estruch R, Ruilope LM, Cosentino F. The year in cardiovascular medicine 2020: epidemiology and prevention. European Heart Journal. 2021;42(8):813-21. https://doi.org/10.1093/eurheartj/ehaa1062">doi: https://doi.org/10.1093/eurheartj/ehaa1062
2. Ralapanawa U, Sivakanesan R. Epidemiology and the Magnitude of Coronary Artery Disease and Acute Coronary Syndrome: A Narrative Review. J Epidemiol Glob Health. 2021;11(2):169-77. doi: https://doi.org/10.2991/jegh.k.201217.001
3. Vieira Lima Aguiar Melão B, Pekala K, Matsoukas K, et al. SIU-ICUD: Epidemiology of Prostate Cancer. Soc Int Urol J. 2025;6(44). doi: https://doi.org/10.3390/siuj6030044
4. Sun L, Parikh RB, Hubbard RA, et al. Assessment and Management of Cardiovascular Risk Factors Among US Veterans With Prostate Cancer. JAMA Netw Open. 2021;4(2):e210070. doi: https://doi.org/10.1001/jamanetworkopen.2021.0070
5. Kuryata O, Sirenko O. The Influence of L-Carnitine in the Complex Treatment of Patients With Renal Dysfunction Who Have Synchronous Chronic Coronary Artery Disease and Prostate Adenocarcinoma. Turkiye Klinikleri J Med Sci. 2019;39(4):381-8. doi: https://doi.org/10.5336/medsci.2019-65452
6. Kuryata OV, Sirenko O, Tykhomyrov A, et al. Plasminogen activator inhibitor-1 and circulating ceruloplasmin levels in men with iron-deficiency anemia and heart failure with concomitant prostate cancer and their dynamics after treatment. Journal of Medical Sciences. 2022;42:72-80. doi: https://doi.org/10.4103/jmedsci.jmedsci_427_20
7. Kim TH, Lim DG, Chung HS, Chang Hwang E, Kang TW, Kwon DD, et al. The prevalence of dyslipidemia in patients with prostate cancer. Prostate Int. 2025;13(3):174-8. doi: https://doi.org/10.1016/j.prnil.2025.03.005
8. Mach F, Koskinas KC, Roeters van Lennep JE, Tokgözoğlu L, Badimon L, Baigent C, et al. 2025 Focused Update of the 2019 ESC/EAS Guidelines for the management of dyslipidaemias: Developed by the task force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS). European Heart Journal. 2025;46(42):4359-78. doi: https://doi.org/10.1093/eurheartj/ehaf190
9. Zhang H, Xu H, Zhang C, et al. Ursodeoxycholic acid suppresses the malignant progression of colorectal cancer through TGR5-YAP axis. Cell Death Discov. 2021;7(1):207. doi: https://doi.org/10.1038/s41420-021-00589-8
10. Lakić B, Škrbić R, Uletilović S, et al. Beneficial Effects of Ursodeoxycholic Acid on Metabolic Parameters and Oxidative Stress in Patients with Type 2 Diabetes Mellitus: A Randomized Double-Blind, Placebo-Controlled Clinical Study. J Diabetes Res. 2024 Feb 29;2024:4187796. doi: https://doi.org/10.1155/2024/4187796
11. Daniels JP, Hernández-Tirado A, Mirocha J, et al. Association between insulin resistance and prostate volume: A 4-year analysis from the Reduction by Dutasteride of Prostate Cancer (REDUCE) Trial. BJUI Compass. 2025;6(9):e70085. doi: https://doi.org/10.1002/bco2.70085
12. Morieri ML, Perrone V, Veronesi C, et al. Improving statin treatment strategies to reduce LDL-cholesterol: factors associated with targets’ attainment in subjects with and without type 2 diabetes. Cardiovasc Diabetol. 2021 Jul 16;20(1):144. doi: https://doi.org/10.1186/s12933-021-01338-y
13. Zeng H, Safratowich BD, Cheng W-H, et al. Ursodeoxycholic Acid Exhibits Greater Inhibitory Effects on Cancerous HCT116 Colon Cells than on Noncancerous NCM460 Colon Cells. Nutrients. 2025;17:1072. doi: https://doi.org/10.3390/nu17061072
14. Levey AS, Stevens LA. Estimating GFR using the CKD Epidemiology Collaboration (CKD-EPI) creatinine equation: more accurate GFR estimates, lower CKD prevalence estimates, and better risk predictions. Am J Kidney Dis. 2010;55(4):622-7. doi: https://doi.org/10.1053/j.ajkd.2010.02.337
15. Martín García A, Mitroi C, Mazón Ramos P, et al. Stratification and management of cardiovascular risk in cancer patients. A consensus document of the SEC, FEC, SEOM, SEOR, SEHH, SEMG, AEEMT, AEEC, and AECC. Rev Esp Cardiol (Engl Ed). 2021;74(5):438-48. doi: https://doi.org/10.1016/j.rec.2020.11.020
16. Khan A, Sarkar E, Chandra A, et al. Serum Insulin, Insulin-Like Growth Factor-1, Testosterone and Lipid Profile Levels in Benign Prostatic Hyperplasia and Prostate Cancer at Diagnosis. Cureus. 2024;16(12):e75342. doi: https://doi.org/10.7759/cureus.75342
17. Zhou Z, Huang Z, Zhao Y, et al. Association between low density lipoprotein cholesterol levels and prostate cancer risk in non-hypertensive middle-aged and older American men. Sci Rep. 2024;14:29096. doi: https://doi.org/10.1038/s41598-024-80190-y
18. Wittert G, Grossmann M. Obesity, type 2 diabetes, and testosterone in ageing men. Rev Endocr Metab Disord. 2022;23(6):1233-42.
doi: https://doi.org/10.1007/s11154-022-09746-5
19. Seo SH, Lee DH, Lee YS, Cho KJ, Park HJ, Lee HW, et al. Co-administration of ursodeoxycholic acid with rosuvastatin/ezetimibe in a non-alcoholic fatty liver disease model. Gastroenterology Report. 2022;10:goac037. doi: https://doi.org/10.1093/gastro/goac037
20. Oostveen RF, Kaiser Y, Hartgers ML, et al. Ursodeoxycholic Acid for Trans Intestinal Cholesterol Excretion Stimulation: A Randomized Placebo Controlled Crossover Study. J Am Heart Assoc. 2024;13(20):e035259. doi: https://doi.org/10.1161/JAHA.124.035259
21. Reiss AB, Vasalani S, Albert J, et al. The Effect of Androgen Deprivation Therapy on the Cardiovascular System in Advanced Prostate Cancer. Medicina. 2024;60:1727. doi: https://doi.org/10.3390/medicina60111727
22. Chou E, Legasto CS, Chin AK, et al. Statin Use in Patients With Cancer: Drug Interaction and Statin Usage. JACC Adv. 2025;4(11 Pt 1):102259. doi: https://doi.org/10.1016/j.jacadv.2025.102259
23. Xia X, Niu H, Xu C, Liu X, Zhang G, Ling J. Revisiting the metabolic crosstalk between type 2 diabetes and hyperuricemia: Pathophysiological insights and therapeutic perspectives. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease. 2026;1872(2):168102. doi: https://doi.org/10.1016/j.bbadis.2025.168102
24. de Jesus M, Mohammed T, Singh M, et al. Etiology and Management of Dyslipidemia in Patients With Cancer. Front Cardiovasc Med. 2022;25(9):892335. doi: https://doi.org/10.3389/fcvm.2022.892335
25. Nissen SE, Lincoff AM, Brennan D, et al. Bempedoic Acid and Cardiovascular Outcomes in Statin-Intolerant Patients. N Engl J Med. 2023;388(15):1353-64. doi: https://doi.org/10.1056/NEJMoa2215024
26. Velez BC, Petrella CP, DiSalvo KH, et al. Combined inhibition of ACLY and CDK4/6 reduces cancer cell growth and invasion. Oncol Rep. 2023;49(2):32. doi: https://doi.org/10.3892/or.2022.8469
27. Makhanets L, Vinnychuk O, Hryhorkiv M. [Statistics: Laboratory Workshop in STATISTICA 12: Tutorial]. Chernivtsi: Chernivets nats un-t im. Yu. Fedkovycha; 2023. 161 р. Ukrainian.
28. Pancholia AK, Kabra NK, Gupta R. Laboratory evaluation of lipid parameters in clinical practice. Indian Heart J. 2024;76(Suppl 1):S29-S32. doi: https://doi.org/10.1016/j.ihj.2024.02.002
29. Gastaldelli A. Measuring and estimating insulin resistance in clinical and research settings. Obesity (Silver Spring). 2022;30(8):1549-63. doi: https://doi.org/10.1002/oby.23503
30. Kwok R, Kishore K, Zafari T, et al. Comparative performance of CKD-EPI equations in people with diabetes: An international pooled analysis of individual participant data. Diabetes Res Clin Pract. 2025;223:112104. doi: https://doi.org/10.1016/j.diabres.2025.112104


 UK
UK  EN
EN