Key words: esophageal leiomyoma, postoperative complications, thoracoscopic surgery, enucleation; minimally invasive surgery, benign esophageal tumors, anastomotic leak, endoscopic vacuum therapy
Ключові слова: лейоміома стравоходу, післяопераційні ускладнення, торакоскопічна хірургія, енуклеація, малоінвазивна хірургія, доброякісні пухлини стравоходу, анастомотична негерметичність, ендоскопічна вакуум-терапія
Abstract
Esophageal leiomyomas are the most common benign tumors of the esophagus. While surgical resection is the definitive treatment for symptomatic cases, it carries a risk of postoperative complications. This study presents a case series on the surgical management of esophageal leiomyomas, with a particular emphasis on postoperative complications and their management. This retrospective case series, conducted at the National Cancer Institute (Kyiv, Ukraine), analyzed patients who underwent surgical treatment for symptomatic esophageal leiomyoma between 2021 and 2024 and developed postoperative complications classified as Clavien-Dindo grade III or higher. Data were collected from medical records, including demographics, tumor characteristics, surgical details, postoperative course, complications, management strategies, and outcomes. The total number of observations was n=3. Three cases with Clavien-Dindo III-IV postoperative complications were analyzed. Complications included anastomotic leakage following Ivor Lewis esophagectomy (managed with endoscopic stenting and endoscopic vacuum therapy – EVT), esophageal perforation during enucleation leading to mediastinitis (managed with stenting and endoscopic vacuum therapy), and a complex case involving hemopericardium, post-cardiac arrest syndrome, peritonitis and diaphragmatic herniation requiring multiple reinterventions. Despite the severity of these complications, multidisciplinary management led to complete recovery of all patients. Histopathological evaluation confirmed benign leiomyoma in all cases. Surgical management of esophageal leiomyomas, particularly for large tumors, is often associated with significant postoperative complications. While minimally invasive techniques may reduce these risks, they do not eliminate them entirely. Endoscopic therapy plays a critical role in the non-surgical management of anastomotic leakage and esophageal perforation. Vigilant postoperative monitoring and a multidisciplinary approach are essential for achieving favorable outcomes in these complex cases.
Реферат
Ускладнення хірургічного лікування лейоміом стравоходу: серія випадків. Козак Є., Кондрацький Ю., Насташенко І., Добржанський О., Пепенін М., Колесник А., Шудрак Є., Городецький А., Турчак В., Коваль Н., Свічкар Я., Українець І., Бліжнікова С. Лейоміоми стравоходу є найпоширенішими доброякісними пухлинами стравоходу. Головним методом лікування симптомних випадків є хірургічне втручання, однак воно пов’язане з ризиком післяопераційних ускладнень. У цій роботі подано серію клінічних випадків хірургічного лікування лейоміом стравоходу з особливим акцентом на післяопераційні ускладнення та їх ведення. Ретроспективна серія випадків, проведена в Національному інституті раку (Київ, Україна), включала пацієнтів, яким у 2021-2024 роках виконано хірургічне лікування симптомної лейоміоми стравоходу та в яких розвинулися післяопераційні ускладнення класу Clavien–Dindo III або вище. Дані отримували з медичної документації; вони включали характеристику пухлини, деталі операції, перебіг післяопераційного періоду, ускладнення, стратегії лікування та результати. Загальна кількість спостережень – n=3. Проаналізовано три випадки з післяопераційними ускладненнями Clavien-Dindo III-IV. Серед ускладнень відзначено: неспроможність анастомозу після операції Льюїса (ендоскопічне стентування та ендоскопічна ВАК-терапія), перфорацію стравоходу під час енуклеації з розвитком медіастиніту (стентування та ендоскопічна ВАК-терапія), а також складний випадок із розвитком гемоперикарда, синдромом після зупинки серця, перитонітом і діафрагмальною грижею, що потребував багаторазових повторних втручань. Попри тяжкість ускладнень, мультидисциплінарний підхід до лікування забезпечив повне одужання всіх пацієнтів. Гістологічне дослідження у всіх випадках підтвердило доброякісну лейоміому. Хірургічне лікування лейоміом стравоходу, особливо великих пухлин, часто супроводжується значними післяопераційними ускладненнями. Мінімально інвазивні методики можуть зменшувати ці ризики, але не усувають їх повністю. Ендоскопічні методи (зокрема стентування та ендоскопічна ВАК-терапія) відіграють ключову роль у консервативному веденні неспроможності анастомозу та перфорації стравоходу. Пильний післяопераційний моніторинг і мультидисциплінарний підхід є визначальними для досягнення сприятливих результатів у таких складних клінічних ситуаціях.
Esophageal leiomyomas are the most common benign tumors of the esophagus, arising from the smooth muscle cells of the esophageal wall. Although the true incidence remains uncertain due to their frequently asymptomatic nature and incidental detection, these lesions are rare, accounting for less than 1% of all esophageal neoplasms. Typically slow-growing and having low malignant potential, esophageal leiomyomas are predominantly submucosal and may occur at any site along the esophagus, with a predilection for the lower two-thirds, corresponding to the greater abundance of smooth muscle tissue in this region [1].
Aim – to characterize the spectrum, timing, and severity (Clavien–Dindo grade III–IV) of postoperative complications following surgical management of symptomatic esophageal leiomyoma at the National Cancer Institute (Kyiv, 2021-2024), and to delineate the diagnostic and therapeutic algorithms employed, with particular emphasis on the role and effectiveness of endoscopic strategies – self-expanding stent placement and endoscopic vacuum therapy (EVT) – in organ-preserving management of anastomotic leakage and esophageal perforation.
Advancements in diagnostic imaging, particularly the increasing use of endoscopy, endoscopic ultrasonography, and cross-sectional imaging modalities, have led to more frequent identification of these tumors [2, 3]. A barium contrast study remains a valuable, noninvasive initial investigation, while endoscopic ultrasonography has emerged as a critical modality for evaluating the intramural characteristics of the tumor and assessing for associated lymphadenopathy [4].
Surgical resection remains the definitive treatment for symptomatic tumors, with the primary objective being complete excision while preserving esophageal integrity and function [5]. Traditional open thoracotomy has been largely supplanted by minimally invasive approaches, particularly thoracoscopic surgery, which offers the advantages of reduced postoperative morbidity, shorter hospital stays, and diminished wound complications [6, 7, 8]. Recently, endoscopic techniques such as submucosal dissection and enucleation have gained prominence, particularly for smaller tumors [9, 10, 11].
Histopathological evaluation characteristically reveals well-circumscribed lesions composed of intersecting fascicles of spindle cells with abundant cytoplasm. Immunohistochemical staining typically demonstrates positivity for desmin and alpha-smooth muscle actin, while typically negative for CD34, CD117, and S100, aiding in the differentiation from gastrointestinal stromal tumors [12]. Although many esophageal leiomyomas are asymptomatic, larger tumors may present with symptoms such as dysphagia, chest pain, retrosternal discomfort, heartburn, and regurgitation, occasionally mimicking malignant esophageal lesions and posing diagnostic challenges [13].
This case series describes our experience in the surgical management of esophageal leiomyomas, with a specific focus on the complications encountered and the strategies employed for their resolution. Despite the generally favorable prognosis associated with esophageal leiomyomas, surgical intervention carries a risk of complications, including esophageal perforation and anastomotic leak, necessitating careful perioperative planning and management.
MATERIALS AND METHODS OF RESEARCH
This is a retrospective case series conducted at the National Cancer Institute, Kyiv, Ukraine. The analysis included patients who underwent surgical treatment for esophageal leiomyoma between 2021 and 2024. Diagnosis was established based on clinical, radiological, endoscopic, and histopathological evaluations. We included patients with symptomatic esophageal leiomyoma confirmed by imaging who underwent surgical resection (either excision or enucleation), with subsequent histopathological confirmation of leiomyoma, and who experienced Clavien–Dindo grade III-IV complications.
Preoperative diagnostic evaluation included esophagogastroduodenoscopy (EGD), endoscopic ultrasonography (EUS), and contrast-enhanced computed tomography (CT) of the chest and abdomen. EUS-guided fine-needle aspiration biopsy was performed to facilitate diagnosis. Immunohistochemical staining was utilized to differentiate leiomyomas from gastrointestinal stromal tumors (GISTs) and other mesenchymal neoplasms. The choice of surgical approach was individualized based on tumor size, location, and involvement of surrounding structures. Data were retrospectively extracted from medical records, including patient demographics, comorbidities, tumor characteristics, surgical details, postoperative complications, and management strategies. The total number of observations was n=3. Statistical analysis: only descriptive statistics were applied. Continuous variables are presented as median and range (min–max); categorical variables as counts and percentages. No inferential tests were performed. No software was used.
We graded postoperative complications using the Clavien-Dindo classification and applied ECCG consensus definitions for esophagectomy-related complications. Esophageal leaks/perforations were managed with endoscopic vacuum therapy (EVT) following published techniques. The Ivor Lewis approach is cited as originally described. Reporting follows the PROCESS guidelines for surgical case series.
The study protocol was reviewed and approved by the Bioethics Commission of the National Cancer Institute, Kyiv, Ukraine (Protocol No. 1347, Meeting date: 03.07.2025). The study complied with the Declaration of Helsinki (2013 revision). All data were de-identified prior to analysis.
The requirement for individual informed consent was waived by the Bioethics Commission due to the retrospective design, use of de-identified data, and minimal risk to participants (Protocol No. 1347, 03.07.2025).
Case presentation
Case 1.
A 38-year-old Caucasian male was admitted with complaints of dysphagia, a globus sensation in the throat, intermittent heartburn, and periodic right upper quadrant abdominal pain. He reported symptoms persisting for several months without prior treatment.
Comorbidities included stage II arterial hypertension, chronic heart disease (stage I chronic heart failure). A congenital urinary tract anomaly-specifically, incomplete duplication of the left renal calyceal-pelvic system – as also noted.
Specific diagnostic procedures
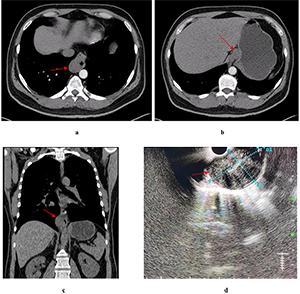
Endoscopic examination revealed a rounded lesion measuring up to 3.5 cm in greatest dimension with an unaltered overlying mucosa. The tumor was located in the lower third of the esophagus and extended into the cardia of the stomach along the posterior wall.
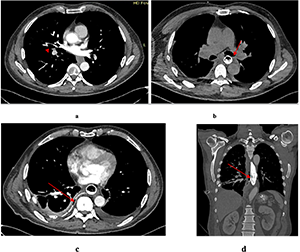
Chest, abdomen, and pelvis CT scan with intravenous enhancement demonstrated a mass originating from the esophageal wall (Fig. 1).
Endoscopic ultrasound (EUS) visualized a hypoechoic lesion originating from the muscular layer of the esophageal wall. The lesion measured 18 mm in basal diameter, extended caudally along the gastric wall, and had a maximum length of 73 mm. It displayed a bean-shaped contour, homogeneous echotexture, and well-defined margins. Fine-needle aspiration biopsy was performed, but did not yield a definitive diagnosis.
Treatment
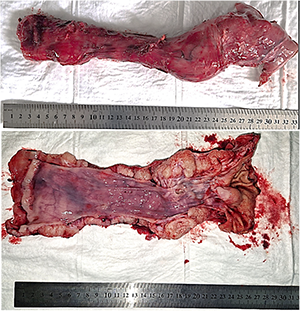
Due to the size and extent of the lesion, surgical enucleation was deemed technically infeasible. A two-stage treatment strategy was employed. The first stage involved diagnostic laparoscopy with biopsy of an enlarged perigastric lymph node and ligation of the left gastric artery. No histological evidence of malignancy was found. Subsequently, a minimally invasive Ivor Lewis esophagectomy and feeding jejunostomy placement were performed.
Postoperative complications
The patient was transferred from the ICU to a specialized surgical ward on postoperative day (POD) 1, after a chest X-ray confirmed no complications. Postoperative care included multimodal analgesia with epidural analgesia, antibiotic therapy (cefazolin, levofloxacin, and metronidazole), antifungal therapy (fluconazole), stimulation of gastrointestinal motility, jejunal feeding, and supplementary parenteral nutrition for 48 hours.
On POD 3, the patient developed fever, and subsequent CT revealed bilateral pneumonia and left-sided hydrothorax, prompting modification of the antibiotic regimen.
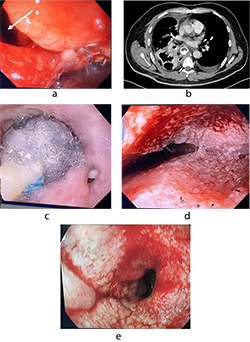
By POD 6, persistent fever has necessitated transition to broad-spectrum antibiotic therapy with colistimethate sodium and meropenem trihydrate. Nil by mouth was initiated with the switch to parenteral nutrition. An anastomotic leak at the esophagogastric junction was diagnosed on POD 7 via endoscopy, prompting stent placement. Initially, satisfactory stent placement was confirmed by CT. On POD 11, ultrasound detected pleural effusion, which was drained. On POD 13, due to persistent fever, the stent was removed. Further endoscopy revealed a defect involving one-third of the anastomotic circumference. CT demonstrated persistent dehiscence (6 mm) along the posterior wall and a second 4 mm defect, with bilateral hydrothorax and right-sided empyema. On POD 23, endoscopic vacuum therapy (EVT) was initiated under general anesthesia (Fig. 2). The EVT system was replaced ten times over the subsequent six weeks, resulting in gradual improvement. CT and endoscopy confirmed gradual improvement. A final endoscopy showed complete closure of the anastomotic defect with no evidence of passage obstruction. The patient was discharged in satisfactory condition on POD 41 for outpatient follow-up.
Complications included an anastomotic leak at the esophagogastric junction (Clavien-Dindo grade IIIb), bilateral lower lobe hospital-acquired pneumonia, external esophagocutaneous fistula, and mediastinitis.
Pathology: histopathological examination revealed a tumor with focal necrosis. Morphological features were most consistent with a gastrointestinal stromal tumor (GIST); however, immunohistochemical (IHC) analysis was required for confirmation. The resection margins were clear of tumor involvement, and all twelve regional lymph nodes were negative for malignancy. IHC demonstrated strong positivity for smooth muscle actin and desmin, supporting a smooth muscle origin. Ki-67 antigen expression was less than 1%. Markers for neurogenic differentiation (S-100) and gastrointestinal stromal tumors (CD34) were negative. In conclusion, the lesion was identified as a large benign leiomyoma of the esophagus.
Case 2.
A 24-year-old Caucasian female was admitted with Grade 4 dysphagia, postprandial fullness, weight loss, and moderate general weakness. She had been experiencing symptoms for six months and had previously sought medical attention for nausea, vomiting, chest pain, and weakness. The patient received inpatient care at her local healthcare facility.
Comorbidities included chronic cholecystitis.
Specific diagnostic procedures
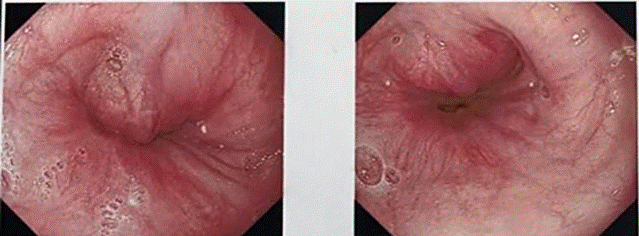
Endoscopic examination revealed a rounded lesion externally compressing an 8 cm segment in the lower third of the esophagus along the right lateral wall, with intact mucosa. Erosions measuring 3-5 mm with semicircular distribution were observed at the gastroesophageal junction, accompanied by reactive hyperplasia of the gastric mucosa (Fig. 3).
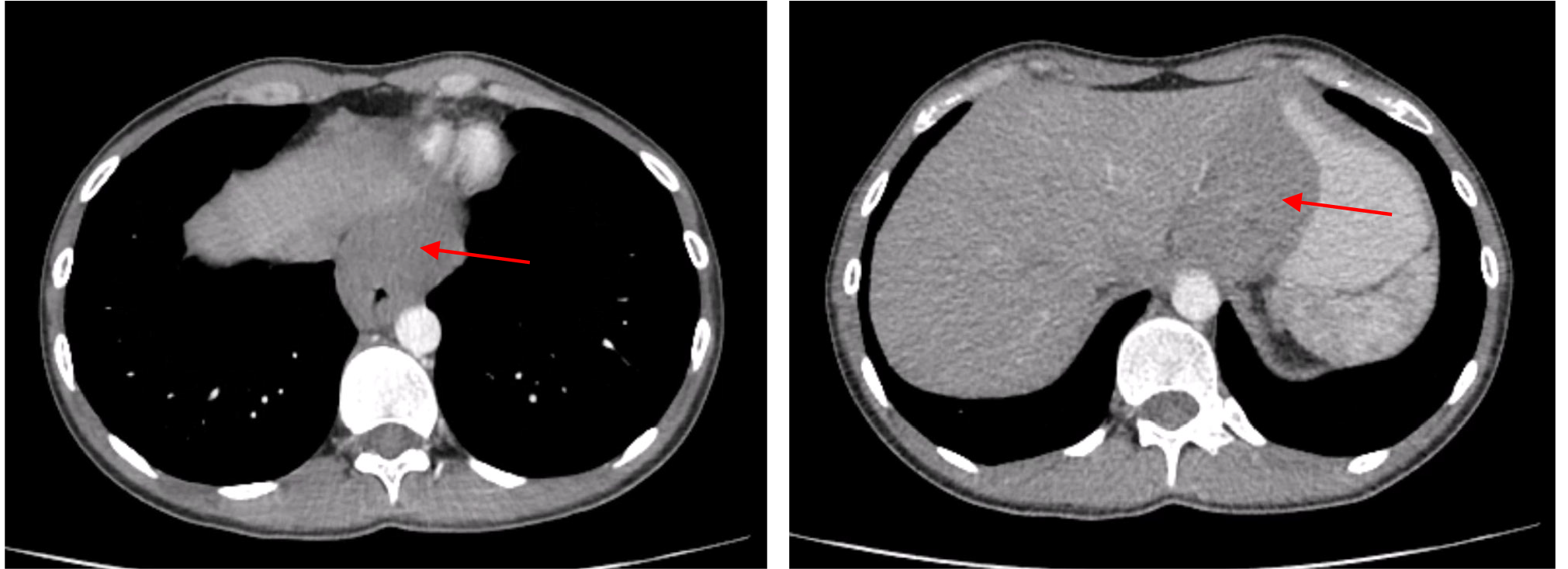
Contrast-enhanced CT of the chest, abdomen, and pelvis showed circumferential thickening and distortion of the abdominal esophagus and gastric cardia, with a mass measuring 48×78 mm and a density of 60-63 Hounsfield units (Fig. 4). perigastric lymph nodes were not radiologically visible. Isolated para-aortic lymph nodes up to 13 mm were noted.
Endoscopic ultrasound (EUS) revealed an irregular, predominantly hypoechoic submucosal lesion measuring approximately 48×48 mm and extending over 80 mm in length in the lower esophagus. The lesion had clear contours, heterogeneous parenchyma, and multiple vascular loci by Doppler imaging, originating from the muscular layer without mucosal invasion. Transesophageal fine-needle biopsy (FNB) was consistent with leiomyoma.
Treatment.
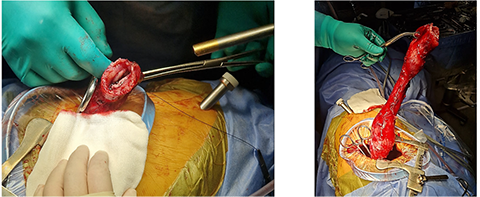
Due to lesion size and extent, surgical enucleation was technically unfeasible. Minimally invasive Ivor Lewis esophagectomy with jejunostomy feeding tube placement was performed (Fig. 5).
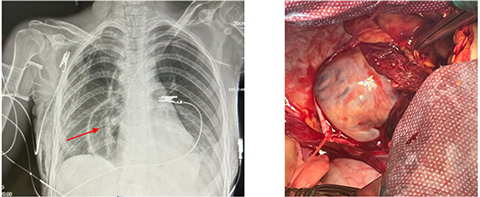
On POD 1, chest radiography revealed a pericardial effusion, necessitating pericardial drainage (Fig. 6a). Subsequently, the patient developed acute clinical deterioration due to hemopericardium, a complication of the drainage procedure, resulting in asystole. Cardiopulmonary resuscitation was performed, successfully restoring spontaneous circulation. The patient's condition rapidly declined, characterized by hypotension, cardiovascular and respiratory failure, and severe chest pain. Contrast-enhanced computed tomography following drainage revealed hydropericardium with imaging suggestive of Budd-Chiari syndrome.
A massive transfusion protocol was initiated. Emergency surgical intervention was performed, consisting of thoracotomy, pericardiotomy, hemostasis via suture ligation of the marginal coronary vein, pericardial fenestration, and drainage of the right pleural cavity (Fig. 6 b). On postoperative day (POD) 6, tracheostomy was performed due to prolonged mechanical ventilation. By POD 14, the patient demonstrated clinical signs of peritonitis with localized guarding in the right iliac fossa, prompting urgent diagnostic laparoscopy with abdominal and pelvic cavity drainage and sanitation. On POD 19, computed tomography revealed herniation of the colon into the left pleural cavity through the esophageal hiatus. Emergency re-laparoscopy was undertaken, including repositioning of the colon into the abdominal cavity, crurorrhaphy, and comprehensive sanitation with drainage. The patient’s condition gradually improved, with resolution of multiple organ dysfunction syndrome (MODS) and hepatic dysfunction. Bedside mobilization commenced, supported by comprehensive care in the intensive care unit (ICU), including mechanical ventilation, renal replacement therapy, intravenous fluid administration, and pharmacologic management. The patient was transferred from the ICU to the surgical ward on POD 26, began oral nutrition, and was discharged in stable condition with outpatient follow-up on POD 34.
Complications: multiple organ dysfunction syndrome (MODS) involving cardiovascular, respiratory, hepatic, and renal failure; post–cardiac arrest syndrome; hemopericardium; intrapericardial hematoma; superior and inferior vena cava syndrome; diaphragmatic herniation; colon translocation into the left pleural cavity; peritonitis; postoperative acute pancreatitis; acute kidney injury; ischemic hepatitis; internal jugular and subclavian vein thrombosis; post-pulmonary embolism syndrome; thoracic stomach syndrome (TSS); and bilateral hospital-acquired pneumonia.
Pathology: histopathological examination confirmed a benign leiomyoma of the esophagus. All ten regional lymph nodes examined were negative for tumor involvement (Fig. 7).
Case 3.
A 55-year-old Caucasian male was admitted with complaints of heartburn and persistent epigastric pain unresponsive to treatment with omeprazole and ranitidine. The patient reported experiencing symptoms for ten months.
The patient’s comorbidities included stage I, grade I essential hypertension (moderate cardiovascular risk) and nodular goiter in a euthyroid state.
Specific diagnostic procedures
Endoscopic examination revealed a polypoid, elastic, mobile lesion approximately 3 cm in diameter, located 32 cm from the incisors. The overlying mucosa appeared intact.
A barium swallow demonstrated a round filling defect on the posterior wall of the lower third of the esophagus, measuring 39×29 mm, with well-defined margins and intramural calcifications. Contrast-enhanced CT of the chest, abdomen, and pelvis showed a well-circumscribed lesion measuring 25×22×30 mm in the middle third of the esophagus, narrowing the esophageal lumen to a slit-like configuration. The external contour of the esophageal wall remained intact (Fig. 8a).
Treatment
Due to its size and location, enucleation was deemed technically feasible. Thoracoscopic enucleation of the esophageal leiomyoma was performed. A mucosal defect occurred at the site of the prior endoscopic biopsy and was closed with interrupted sutures. The right pleural cavity was drained via the seventh intercostal space.
Postoperative complications
On postoperative day (POD) 1, the patient was diagnosed with esophageal perforation complicated by mediastinitis. Leakage was confirmed by assessing drain output after oral administration of a contrast dye. CT imaging of the chest, abdomen, and pelvis revealed signs of esophageal wall perforation. Endoscopic clipping and esophageal stent placement were performed, and a nasojejunal feeding tube was inserted for enteral nutrition. On POD 6, fluid collections in both pleural cavities were drained under ultrasound guidance. Follow-up endoscopy on POD 7 revealed a persistent esophageal wall defect with dense fibrin deposition above the stent. By POD 10, granulation tissue formation was observed at the defect site. On POD 18, CT imaging revealed an isolated esophago-atmospheric fistula extending to a drainage tract through the sixth intercostal space on the right. The stent was removed, and the esophageal defect had decreased to 7 mm. Endoscopic vacuum therapy (EVT) was initiated under general anesthesia. On POD 21, the EVT system was replaced and subsequently removed on POD 23. Fistulography was performed, followed by thrice-daily irrigation of the fistulous tract with antiseptic solutions. Daily drainage from the mediastinum amounted to approximately 85 mL of serous, saliva-mixed exudate. The drain was connected to a pleural aspirator. The patient received enteral feeding with 1000 mL of nutritional formula and 1500 mL of water per day. At discharge on POD 64, the patient was in satisfactory condition. He was afebrile, communicative, and reported no significant complaints. Sutures had been removed, and surgical drains remained in place and functional. The patient was transferred to a military hospital for continuation of care until complete closure of the esophago-atmospheric fistula.
Complications included esophageal perforation, bilateral lower-lobe hospital-acquired pneumonia, external esophagocutaneous fistula, and mediastinitis.
Pathology: a benign esophageal leiomyoma was identified, measuring 3.0×2.0×1.5 cm.
RESULTS AND DISCUSSION
Esophageal leiomyomas are rare benign tumors that originate from the smooth muscle layer of the esophageal wall. Although they generally follow an indolent course and are associated with an excellent prognosis after surgical resection, their management may be complicated, particularly in cases involving large tumors or those located near critical anatomical structures. In this case series, we reported three patients who underwent surgical treatment for esophageal leiomyomas, each of whom experienced significant postoperative complications that required active clinical management.
Minimally invasive approaches, such as thoracoscopic surgery and laparoscopic-assisted esophagectomy, have largely supplanted open thoracotomy in the treatment of esophageal leiomyomas. These techniques are favored due to reduced postoperative pain, lower rates of pulmonary complications, and shorter hospital stays. However, as demonstrated in this series, these benefits do not entirely eliminate the risk of serious complications.
The most critical complication observed was anastomotic leakage, which occurred in two patients following Ivor Lewis esophagectomy. Anastomotic leakage is a serious and potentially life-threatening postoperative complication that can lead to prolonged hospitalization, increased morbidity, and the need for additional interventions (Table). In both cases, endoscopic management – including esophageal stenting and endoscopic vacuum therapy (EVT) – was successfully employed to control the leaks non-operatively. EVT, in particular, proved highly effective in promoting defect closure and controlling mediastinal contamination, consistent with current literature supporting its utility in anastomotic leak management [14].
Another major complication was postoperative mediastinitis secondary to esophageal perforation – an infrequent but dangerous adverse event following enucleation. Prompt diagnosis and a multimodal approach incorporating endoscopic stenting, drainage, and EVT were critical for successful patient recovery.
One patient in our series developed an unusually complex combination of complications, including hemopericardium, post-cardiac arrest syndrome, peritonitis, and diaphragmatic herniation with translocation of the colon into the pleural cavity. This case required multiple surgical interventions and comprehensive multidisciplinary care. Despite the severity of these complications, the patient ultimately recovered, emphasizing the importance of vigilant monitoring and timely intervention for delayed or progressive postoperative issues.
Esophageal leiomyoma is the most common benign esophageal tumor; for symptomatic disease, enucleation remains standard, and minimally invasive approaches reduce peri-operative morbidity in contemporary series [15]. For large or unfavorably located tumors, conversion to esophagectomy may be required when safe dissection is not feasible or malignancy cannot be excluded. In our single-center series (n=3), major complications included an intrathoracic anastomotic leak after Ivor Lewis esophagectomy; reported leak incidences vary widely (~3-30%), emphasizing the need for standardized definitions [16]. We therefore graded complications with the Clavien-Dindo system and applied ECCG consensus definitions to enhance comparability [17, 18]. Management relied on self-expanding stents and endoscopic vacuum therapy (EVT); meta-analytic and comparative data show high EVT success and, in several analyses, superiority to stents, supporting its use alone or in hybrid strategies [19, 20]. Complete recovery in all cases underscores the value of early detection, timely endoscopic rescue, source control/drainage, nutritional support, and coordinated multidisciplinary care.
Case number Postoperative complications (Clavien-Dindo Grade) Case 1 Anastomotic leak (Grade IIIb), bilateral lower lobe hospital-acquired pneumonia (Grade II), esophagocutaneous fistula (Grade IIIb), mediastinitis (Grade II). Case 2 Hemopericardium (Grade IVa), cardiac arrest syndrome (Grade IVa), peritonitis (Grade IIIb), postoperative acute pancreatitis (Grade II), MODS (cardiovascular failure, respiratory failure, liver failure, renal failure) (Grade IVb), PE (Grade II), translocation of the colon into the left pleural cavity (Grade IIIb), hospital-acquired bilateral pneumonia (Grade II). Case 3 Esophageal perforation (Grade IIIb), pneumonia (Grade II), esophagocutaneous fistula (Grade IIIb), mediastinitis (Grade II).
Our findings highlight the increasing complexity of surgical management as tumor size and esophageal wall involvement grow. While enucleation remains the preferred treatment for small, well-circumscribed tumors, larger or more invasive leiomyomas may necessitate esophagectomy – an approach associated with higher perioperative risks.
Histopathological analysis in all cases confirmed the diagnosis of benign leiomyoma. Immunohistochemistry played a key role in differentiating these lesions from gastrointestinal stromal tumors (GISTs), which require distinct therapeutic strategies.
The outcomes in our patients underscore that while minimally invasive surgical techniques offer significant benefits, they require a high level of technical skill and meticulous perioperative planning to avoid complications. Additionally, the integration of endoscopic modalities into postoperative care has greatly enhanced the management of complications such as leaks and fistulas, contributing to improved patient outcomes [22, 23].
CONCLUSION
This study is limited by the small sample size, which reflects the rarity of symptomatic esophageal leiomyoma, and by its retrospective design. Nonetheless, the cases presented offer valuable insight into the complexities of surgical decision-making and complication management in this patient population.
Contributors:
Kozak Ye., Kondratskyi Yu., Nastashenko I., Dobrzhanskyi O., Pepenin M., Kolesnyk A., Shudrak Ye., Horodetskyi A., Turchak V., Koval N., Svichkar Ya., Ukrainets I., Blizhnikova S. – contributed to the retrospective data collection, statistical analysis and manuscript writing. Each author has reviewed and approved the final version of the manuscript.
Funding. This research received no external funding.
Conflict of interests. The authors declare no conflict of interest.
REFERENCES
1. Gowrie S, Noel A, Wooten C, Powel J, Gielecki J, Zurada A, et al. Slicing Through the Options: A Systematic Review of Esophageal Leiomyoma Management. Cureus. 2025;17(4):e81614. doi: https://doi.org/10.7759/cureus.81614
2. Impellizzeri G, Donato G, De Angelis C, Pagano N. Diagnostic Endoscopic Ultrasound (EUS) of the Luminal Gastrointestinal Tract. Diagnostics (Basel). 2024;14(10):996. doi: https://doi.org/10.3390/diagnostics14100996
3. Asaf BB, Bishnoi S, Puri HV, et al. Robotic enucleation of oesophageal leiomyoma: technique and surgical outcomes. J Minim Access Surg. 2022;18(1):84-9. doi: https://doi.org/10.4103/jmas.JMAS_263_20
4. Li W, Shao M, Hu S, Xie S, He B. The diagnostic value of endoscopic ultrasound for esophageal subepithelial lesions: a review. Medicine (Baltimore). 2024;103(46):e40419. doi: https://doi.org/10.1097/MD.0000000000040419
5. Tien TPD, Trung LV, Nguyen HN. Minimally Invasive Enucleation of Esophageal Leiomyoma: Experience From a Single Institution. Cureus. 2024;16(10):e71384. doi: https://doi.org/10.7759/cureus.71384
6. A-Lai GH, Hu JR, Yao P, Lin YD. Surgical Treatment for Esophageal Leiomyoma: 13 Years of Experience in a High-Volume Tertiary Hospital. Frontiers in Oncology. 2022;12:876277. doi: https://doi.org/10.3389/fonc.2022.876277
7. Mishra S, Jain N, Soni B, et al. Thoracoscopic enucleation of oesophageal submucosal tumours in prone position gives excellent long-term outcome: A single-centre experience. J Minim Access Surg. 2022;18(3):401-7. doi: https://doi.org/10.4103/jmas.JMAS_169_21
8. Tun KM, Dhindsa BS, Dossaji Z, et al. Efficacy and safety of submucosal tunneling endoscopic resection…: a systematic review and meta-analysis of 2900 patients. iGIE. 2023;2(4):529-537.e2. doi: https://doi.org/10.1016/j.igie.2023.08.005
9. Liapis SC, Baloyiannis I, Perivoliotis K, et al. Submucosal Tunneling Endoscopic Resection for Upper Gastrointestinal Subepithelial Lesions: A Systematic Review and Meta-Analysis. Journal of Gastrointestinal Cancer. 2025;56:110.
doi: https://doi.org/10.1007/s12029-025-01235-7
10. Kinney T, Waxman I. Treatment of benign esophageal tumors by endoscopic techniques. Semin Thorac Cardiovasc Surg. 2003;15(1):27-34. doi: https://doi.org/10.1016/S1043-0679(03)00004-2
11. Daitch ZE, Heller SJ. Endoscopic ultrasonography in esophageal carcinoma: a narrative review. Annals of Esophagus. 2023;6:14.
doi: https://doi.org/10.21037/aoe-21-25
12. Triantafyllou A, Mela E, Theodoropoulos C, et al. Addressing Anastomotic Leak After Esophagectomy: Insights from a Specialized Unit. J Clin Med. 2025;14(11):3694. doi: https://doi.org/10.3390/jcm14113694
13. Wang R, Guo Y, Duan X, Jiang H. Robot-assisted resection of oesophageal leiomyoma: a single-centre retrospective analysis of 57 cases. ANZ Journal of Surgery. 2025;95(3):356-62. doi: https://doi.org/10.1111/ans.70022
14. Ubels S, Lubbers M, Verstegen MHP, et al. Treatment of anastomotic leak after esophagectomy: insights of an international case vignette survey and expert discussions. Diseases of the Esophagus. 2022;35(12):doac020. doi: https://doi.org/10.1093/dote/doac020
15. Milito P, Asti E, Aiolfi A, et al. Clinical Outcomes of Minimally Invasive Enucleation of Leiomyoma of the Esophagus and Esophagogastric Junction. J Gastrointest Surg. 2020;24(3):499-504. doi: https://doi.org/10.1007/s11605-019-04210-3
16. Ubels S, Verstegen MHP, Rosman C, Reynolds JV. Anastomotic leakage after esophagectomy for esophageal cancer: risk factors and operative treatment. Annals of Esophagus. 2021;4:8. doi: https://doi.org/10.21037/aoe-2020-18
17. Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien–Dindo classification of surgical complications. Annals of Surgery. 2009;250(2):187-96. doi: https://doi.org/10.1097/SLA.0b013e3181b13ca2
18. Low DE, Alderson D, Cecconello I, et al. International consensus on standardization of data collection for complications associated with esophagectomy. Annals of Surgery. 2015;262(2):286-94. doi: https://doi.org/10.1097/SLA.0000000000001098
19. Livingstone I, Maloney A, Chowdhury A, Caddy G. Current Status of Endoscopic Vacuum Therapy in the Management of Esophageal Perforations and Post-Operative Leaks. Clinical Endoscopy. 2021;54(6):789-97. doi: https://doi.org/10.5946/ce.2021.240
20. Han S, Girotra M, Abdi M, et al. Endoscopic vacuum therapy. iGIE. 2024;3(3):333-41. doi: https://doi.org/10.1016/j.igie.2024.06.003
21. Papadakos SP, Argyrou A, Katsaros I, et al. The Impact of EndoVAC in Addressing Post-Esophagectomy Anastomotic Leak in Esophageal Cancer Management. Journal of Clinical Medicine. 2024;13(23):7113. doi: https://doi.org/10.3390/jcm13237113
22. Goenka MK, Goenka U. Endotherapy of leaks and fistula. World Journal of Gastrointestinal Endoscopy. 2015;7(7):702-13. doi: https://doi.org/10.4253/wjge.v7.i7.702
23. Vohra I, Gopakumar H, Sharma NR, Puli SR. Efficacy of endoscopic vacuum therapy in esophageal luminal defects: systematic review and meta-analysis. Clinical Endoscopy. 2025;58(1):53-62. doi: https://doi.org/10.5946/ce.2023.282


 UK
UK  EN
EN