Key words: creatine kinase, physical exercise, physical fitness, muscle damage
Ключові слова: креатинкіназа, фізичні вправи, фізична форма, пошкодження м’язів
Abstract
Creatine kinase (CK) is a key enzyme in cellular energy metabolism, playing a role in the regeneration of adenosine triphosphate through the phosphocreatine system, particularly in tissues with high energy demands such as skeletal muscle. Under normal conditions, CK is present within muscle cells, so its blood levels are relatively low. However, strenuous or unusual physical exercise especially with eccentric contractions can cause mechanical and metabolic stress that disrupts muscle fiber integrity and increases cell membrane permeability. Microdamage to the sarcolemma, accompanied by oxidative stress, inflammatory responses, and disruption of intracellular calcium homeostasis, are thought to cause the release of CK from muscle cells into the bloodstream. Although CK is widely used as a biomarker of exercise-induced muscle damage, its increased levels do not always correlate directly with the degree of muscle damage or functional impairment, so the exact mechanism of post-exercise CK release remains incompletely understood. The aim of this study was to determine the effect of physical exercise on increasing CK levels. We reviewed several literature databases, including Scopus, Pubmed, Web of Science, and Science Direct, for our systematic review investigation. The search was conducted for articles published between 2015-2025 that discussed physical exercise and CK. Using the Scopus, Web of Science, Pubmed, and Science Direct databases, 1,450 published publications were identified. For this systematic review, nine papers that met the inclusion criteria were selected and reviewed. This study evaluated the standard operating procedure using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). Based on 9 reviewed papers it has been proven that physical exercise has been proven to increase CK levels significantly. Exercise-induced CK elevations serve as a valuable, albeit nonspecific, indicator of muscle damage. Understanding the variability in CK responses is critical to designing training and recovery programs for athletes in order to achieve optimal physical performance.
Реферат
Зміни креатинкінази, викликані фізичними вправами: систематичний огляд. Путро Б.Н., Вібава Дж.К., Аюбі Н. Креатинкіназа (КК) – ключовий фермент клітинного енергетичного метаболізму, що відіграє роль регенерації аденозинтрифосфату через фосфокреатинову систему, особливо в тканинах з високими енергетичними потребами, як-от скелетні м’язи. За нормальних умов КК присутній у м’язових клітинах, тому його рівень у крові відносно низький. Однак інтенсивні або незвичайні фізичні навантаження, особливо з ексцентричними скороченнями, можуть викликати механічний та метаболічний стрес, який порушує цілісність м’язових волокон та збільшує проникність клітинної мембрани. Вважається, що мікроушкодження сарколеми, що супроводжуються окиснювальним стресом, запальними реакціями та порушенням внутрішньоклітинного кальцієвого гомеостазу, призводять до вивільнення КК з м’язових клітин у кровотік. Хоча КК широко використовується як біомаркер пошкодження м’язів, викликаного фізичними вправами, її підвищений рівень не завжди прямо корелює зі ступенем пошкодження м’язів або функціональними порушеннями, тому точний механізм вивільнення КК після тренування залишається недостатньо вивченим. Метою цього дослідження було визначення впливу фізичних вправ підвищення рівня КК. Для проведення систематичного огляду ми проаналізували кілька баз даних наукової літератури, включаючи Scopus, PubMed, Web of Science та Science Direct. Пошук проводився за статтями, опублікованими в період з 2015 до 2025 року, у яких обговорювалися фізичні вправи та КК (креатинкінази). За допомогою баз даних Scopus, Web of Science, PubMed та Science Direct було виявлено 1450 опублікованих робіт. Для цього систематичного огляду було відібрано та проаналізовано дев'ять статей, що відповідають критеріям включення. У цьому дослідженні оцінювалася стандартна робоча процедура з використанням PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses). З аналізу 9 статей було доведено, що фізичні вправи значно підвищують рівень КК. Підвищення рівня КК, викликане фізичними вправами, є цінним, хоч і неспецифічним індикатором ушкодження м’язів. Розуміння варіабельності реакції КК має вирішальне значення для розробки тренувальних і відновлювальних програм для спортсменів з досягнення оптимальної фізичної працездатності.
Physical exercise is known as a non-pharmacological therapy in an effort to improve the health and standard of living of humans. However, physical exercise, in addition to making you healthy, also triggers certain conditions that result in an imbalance in the body's homeostasis if not done properly and correctly according to WHO recommendations. A rise in ROS (reactive oxygen species) is another indicator that physical activity causes oxidative stress [1]. Exercise causes the body's oxygen consumption to rise by 20-100 times greater than usual, making it impossible for the oxygen supply to keep up [2]. It is well recognized that the body contains a number of ROS, such as hydrogen peroxide (H2O2), hydroxyl radicals (OH-), and superoxide anions (O2-). When physical exercise causes stress, these ROS are known to be essential for cellular physiological functions [3]. During exercise, skeletal muscles also help produce ROS, which is essential for gene expression, cellular signal transduction, strength enhancement, and muscle mass maintenance. However, excessive ROS levels will interfere with muscle contraction and performance continuity [2]. However, we are all aware that excessive ROS production during exercise can lead to oxidative stress, which exacerbates existing medical issues [3]. The body's overproduction of ROS can potentially damage cell biomacromolecules, leading to aging, muscle cell death, and damage to lipids, proteins, and nucleic acids physiological events that happen during physical activity [4]. Apart from that, ROS is a normal physiological reaction that happens during exercise to initiate additional signal transductions [5].
The main cause of exercise-induced muscle damage (EIMD) is unfamiliar physical activity, particularly if it includes a lot of eccentric contractions [6]. Along with a brief loss of muscle strength, this breakdown process usually causes delayed onset muscle soreness (DOMS), which usually starts a few hours after activity and peaks gradually over a few days [7]. In recent decades, numerous ideas have been proposed to explain the process of deterioration [8], however, the precise process remains unclear. After a great deal of fascial research in recent years, it has been shown that fascia is the main cause of pain during eccentric exercise [9]. Additionally, during DOMS episodes, deep fascial shear dysfunction has been noted, and this is correlated with later DOMS recovery [10]. Therefore, the function of fascial sliding may have a significant impact on DOMS; however, a thorough investigation of this link has not yet been conducted. Additionally, there are biomarkers, including creatine kinase (CK), that indicate muscle injury.
The enzyme creatine kinase (CK), sometimes referred to as creatine phosphokinase, is involved in the synthesis of energy. Tissues that consume a lot of energy, such as the brain and muscles, especially the heart, have higher quantities of this enzyme. Serum CK enzyme levels range from 20-35 to 200-400 U/L at baseline [11]. When exercise damages the skeletal muscle cell structure in the sarcolemma and Z-disc levels, creatine kinase will leak into the circulation [12]. The proportional amount of CK produced, the degree of CK enzyme activity released, and the rate of CK clearance from the blood will all be reflected in the measured serum CK [13]. Additionally, it has been proposed that muscle cells "voluntarily" release adenosine triphosphate (ATP)-consuming enzymes like CK in order to avoid cell death under crucial metabolic conditions [14]. Instead of linking CK to "muscle damage," some studies now associate it with "membrane damage" or "membrane permeability." One qualitative marker of muscle injury, especially in the context of endurance exercise, has been suggested to be elevated plasma CK activity [15]. Serious muscle problems are generally thought to be indicated by serum CK values greater than 5000 U/L [14]. CK has also been proposed as an indicator of recovery, but due to high variability in baseline levels or in response to exercise, there is no general consensus. The underlying mechanism by which exercise can raise CK levels as a sign of muscle injury and the consequences for day-to-day living are still unknown. To accurately explain how CK reacts during exercise, it is crucial to understand its phases and mechanisms. Therefore, the aim of this study was to examine in-depth how physical exercise affects CK levels and to examine the underlying mechanisms.
MATERIALS AND METHODS OF RESEARCH
This study was a systematic review, in which the researchers conducted extensive searches through journal databases such as PubMed, Science Direct, Web of Science, and Scopus. These databases are considered to be the world's largest for collecting high-impact papers with a strong scientific foundation, ensuring their legitimacy and validity are undeniable. Using this first-come, first-served search method, duplicate articles were removed. Using predetermined inclusion and exclusion criteria, the search results were then further refined.
The inclusion criteria for this study were developed reviewing publications since 2015 to 2025 that addressed physical activity and creatine kinase. Additionally, journals that did not adhere to scientific validity requirements or were not listed in reliable search indexes like Scopus, Web of Science, Pubmed, or Science Direct were not included in our research.
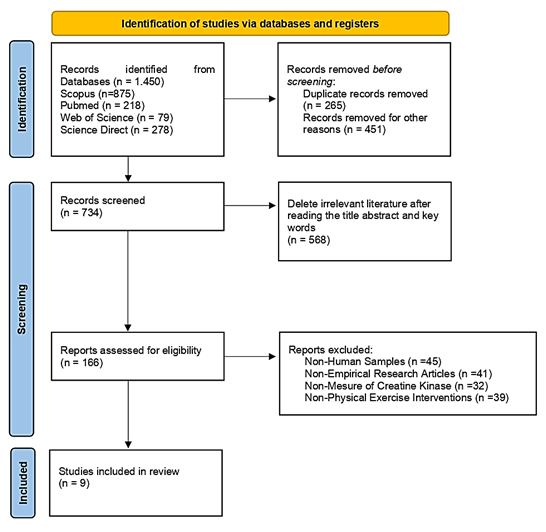
The full texts, abstracts, and titles of the papers were added to the Mendeley database after initial evaluation and confirmation. In the first phase, 1,450 publications were found using the databases Scopus, Science Direct, Pubmed, and Web of Science. After the title appropriateness screening, 734 acceptable papers were selected for the second screening stage (Fig.). Following a review of the title, abstract, and keywords, 166 papers were selected for the third round. Upon reviewing each publication, we determined that they matched the inclusion requirements, which include the following: the study must be original, the parameter must be a creatine kinase biomarker, the intervention must be physical activity of any kind, and the sample must be human. At this point, we organized the items based on their overall suitability. Nine papers that met the inclusion criteria were selected for analysis after a thorough review and observation process. The operational requirement for this study was to comply with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). In addition, all papers have met research ethics and are in accordance with the Helsinki Declaration.
RESULTS AND DISCUSSION
Author Design Participants Participants Age Intervention Outcome (Chen et al., 2025) [6] Randomized controlled trials 24 males 18-25 years Exercise Program Eccentric exercise with The following is the Active range of motion is 30° Participants only do eccentric workouts while seated on the dynamometer Eccentric exercise has been shown Intervention of tissue flotation after eccentric exercise inhibits the increase in creatine kinase (Santos Cerqueira et al., 2021) [16] Randomized controlled trials Thirty healthy young men 18 to 35 years Exercise Program Participants performed A total of 10 sets The volunteer's knees were passively positioned at a 30° angle (0°=total extension) before each contraction. Subjects were instructed Participants were instructed Eccentric training has been shown to significantly increase creatine kinase levels after intervention (Allard et al., 2023) [17] Randomized controlled trials Hundred 62-66 years Exercise Program Participants of the Nijmegen March walk Each day, participants walked 30, 40 and 50 kilometers. For four consecutive days, the exercise intervention was conducted There was a significant increase in CK levels at its peak 2 days after exercise and on the third day CK levels tended to decrease. (Bray et al., 2025) [18] Randomized controlled trials Eleven senior club male fast bowlers 27.3±7.0 years Exercise Program The CA-AIS fast bowling skills test is finished by participants. To put it briefly, bowlers The other players finish a physical exercise that simulates fielding in the interim. There was a significant increase in CK levels immediately after the intervention (D’Alleva et al., 2025) [19] Randomized controlled trials Seventeen participants 40 years Exercise Program Twenty-four weeks Warming up is done before treatment. Following that, runners might choose to run a half, thirty, or full marathon. There was a significant increase in CK levels immediately after the marathon running intervention. (Biss et al., 2023) [20] Randomized controlled trials Twelve healthy participants 18-45 years Exercise Program Stand, squat, biceps curl, toe stand with whole body extension, and other exercises included in the book are examples of electromyostimulation exercises. Plank, deep squat, knee tuck walk, push-up, and other exercises included in the manuscript contents are examples of circuit training Both types of exercise were shown to significantly increase CK levels (Beiter et al., 2025) [21] Randomized controlled trials Eighteen triathletes 32-33 years Exercise Program A one-hour running session on a motorized treadmill (Saturn, h/p/cosmos) was the workout test. After a 10-minute warm-up at the starting pace needed to run at 75% of the individual anaerobic threshold, the activity progressed to a 50-minute main exercise at a steady pace that corresponded to 90% of the individual anaerobic threshold. Normal ambient temperature (18–21°C, N) was used for the first workout test. After 7–14 days, the second training session was conducted in a moderately hot atmosphere with a temperature of 28-30°C There was a significant increase in CK levels in both groups after the exercise intervention both at normal and at high temperature (Kusmierczyk et al., 2024) [22] Randomized controlled trials Twenty-seven participants 19-25 years Exercise Program The treadmill was used for the downhill running exercise, which was started at a speed of 6 km/h on a -10% slope. Participants' speed was modified to reach 60% of their precisely recorded VO2max after four minutes. Cardiorespiratory parameters were continuously assessed for an hour while this speed was maintained There was a significant increase in CK levels after the intervention (Arriel et al., 2018) [23] Randomized controlled trials 28 trained cyclists 25-28 years Exercise Program The maximal incremental cycling test (MICT) Cyclists carried out one of the four interventions listed below right after the MICT: Two 5-minute occlusion/5-minute reperfusion episodes (PEIC or SHAM, 2 x 5) or five 2-minute occlusion/2-minute reperfusion sessions (PEIC or SHAM, 5 x 2) are other options. Unilateral treatments of SHAM (20 mmHg) or PEIC (50 mmHg above systolic blood pressure) were administered to alternate thighs. A second MICT was conducted 24 hours following There was a significant increase in CK levels after the intervention
an isokinetic dynamometer
and eccentric knee flexion exercises, to train the quadriceps and the muscles
of the lower extremities.
exercise's intensity:
10 sets of 12 repetitions,
with a 30-second rest period, for a total of 120 repetitions.
to 90° knee flexion and 60°/s angular velocity.
to significantly increase creatine kinase levels.
120 eccentric movements as closely as possible using the knee extensor muscles.
of 12 repetitions, with 30 seconds rest between sets.
to perform knee extension briefly for dynamometer measurement, and then reduce gradually to 90° knee flexion (60° total range) at a rate
of 60 degrees/second.
to measure the knee movement flow measured by the dynamometer as soon as possible. Each eccentric action lasted for one day, and was performed by the extension patient for one day
participants
at a moderate intensity.
(in pairs) executed four distinct fast bowling periods. Using line (off- and leg-stump) and length (short-, good-, and full-) variations, one player bowled a series of standard bowls
(in 4-over sequence)
at match intensity.
To prolong the bowling spell and meet the over requirements of each experimental trial, the same
4-over routine was repeated
of training were done before the marathon.
was conducted
on the research sample.
the intervention
This study's goal was to ascertain the impact of physical exercise in increasing CK levels after intervention. The results proved that CK levels increased significantly after doing physical exercise. The results of previous studies prove that eccentric workout utilizing an isokinetic dynamometer that involves eccentric knee flexion movements, exercise intensity of a total of 120 repetitions in 10 sets of 12, 30 seconds rest interval proved significant in increasing CK levels after intervention [6]. The findings of additional research also support the notion that the research sample given the intervention to perform 120 maximal eccentric movements with the knee extensor muscles in ten sets of twelve repetitions, with a 30-second break between sets proved significant in increasing CK levels after the intervention [16]. So scientifically, eccentric exercise is proven to increase CK levels. However, we must know how the stages of increase occur and what underlies the increase.
Elderly people who had done previous training so that they participated in the Nijmegen march activities, namely walking with moderate intensity by walking 30, 40, 50 km per day carried out for 4 consecutive days, proved to be significant in increasing CK levels after the exercise intervention [17]. Another finding from the bowling club study was that participants finished a fast bowling skills test in which bowlers (in pairs) executed four distinct fast bowling spells. While the other participant engaged in a simulated fielding exercise, one player executed a series of standardized bowls (in four over sequence) at match intensity, incorporating variations of line (off- and leg-stump) and length (short-, good-, and full-). CK levels significantly increased following the intervention, according to the results [18]. Other data proved that 24 weeks of marathon race preparation training, then participants ran a marathon consisting of running a half, 30 km, and full marathon proved that CK levels increased significantly after the intervention [19].
Supporting this evidence, it is also demonstrated that the findings of additional research support the same conclusion, namely that physical activity intervention raises CK levels [20]. The results of another study conducted on participants with an exercise test intervention for 1 hour on a treadmill and in addition the researchers also compared the effect of temperature on increasing CK levels. The results proved that both the occurrence of temperature differences between cold and hot temperatures after physical exercise interventions proved significant in increasing CK levels [21]. In a different study, participants were given a treadmill with a downhill running exercise intervention on a -10% slope. The participants' initial speed was set at 6 km/h, and after 4 minutes, it was changed to reach 60% of their VO2max, which was precisely measured and maintained for the duration of the intervention. The findings demonstrated that following the physical exercise intervention, CK levels increased [22]. CK levels also increased after the study sample performed the maximal incremental cycling test [23]. So from the results of the review of several studies that have been compiled in this systematic review, all data show the occurrence of CK levels significantly after performing physical exercise interventions. However, it is known to have increased and we must also know the mechanism underlying the increase in CK levels after physical exercise intervention. Therefore, the following discussion will explore in depth how the physiological mechanisms underlying the increase in CK levels after physical exercise.
Physiological mechanisms of physical exercise in increasing creatine kinase levels
Because it is an enzyme of energy and phosphate metabolism involved in the rapid supply and storage of energy, creatine kinase, an intracellular dimeric or octameric protein, is found in body tissues with high energy consumption [24]. One important modulator of cellular energy homeostasis is creatine kinase (CK). CK creates a sizable pool of quickly diffusing phosphocreatine through the reversible interconversion of creatine to phosphocreatine, which buffers ATP levels in the body both geographically and temporally. Therefore, particularly during activity, CK is essential for tissues like muscle and the brain that have high and changing energy demands [24]. This enzyme, which belongs to the phosphoryl transferase family, makes sure that a phosphoryl group is transferred from creatine phosphate to adenosine diphosphate in a reversible manner, creating adenosine triphosphate [25]. In regular laboratory diagnostics, CK has become an essential diagnostic marker because of its subcellular location and variety of functions [26]. In trauma, oxygen deprivation or muscle exhaustion cell homeostasis is upset cause cell damage and cell membranes become leaky. Intracellular components like CK seep out when the membrane ruptures. The subsequent increase in total CK concentration, which can be measured in blood serum, is caused by the sum of all CK isoenzyme fractions, which are primarily supplied by the skeletal muscle isoenzyme fraction. This may be several times higher than typical levels [12].
A substantial intracellular phosphocreatine pool that prevents a precipitous decline in the global ATP concentration and acts as an efficient temporal energy buffer, is formed in part by isoenzymes. The idea of CK isoenzyme subcellular compartmentation and the restricted free passage of even smaller molecules within the cell, like adenine nucleotides, serve as the foundation for additional CK functions [27]. Despite their partial solubility, CK isoenzymes do exhibit some degree of subcellular structural interaction. Metabolite channeling, also referred to as functional coupling, is the process by which CK is directly or indirectly associated with processes that supply or consume ATP, forming microcompartments that often allow direct ADP and ATP exchange between association partners without combining with the bulk solution [28].
For maximum ATP, the local ATP/ADP ratio around the cellular ATPase should be kept high; for oxidative phosphorylation the mitochondrial matrix should have a relatively low ATP/ADP ratio that is possible by the differential microcompartmentalization of CK isoenzymes. According to some theories, CK reactions serve as a kind of intracellular spatial energy transport that makes it easier for high-energy phosphate to move from the mitochondria, which produce ATP, to the cytosol, which uses it, and for the product to return to the mitochondria for rephosphorylation [29]. The CK/phosphocreatine system may theoretically offer the exact localization of mitochondrial and cytosolic isoenzymes, as well as the somewhat quicker diffusion rate of phosphocreatine in comparison to ATP, that results in a spatial "energy shuttle" or "energy circuit," linking the locations of energy generation and consumption [24]. At least in polar and large cells, there is growing evidence that CK and creatine microcompartments and a CK/phosphocreatine shuttle exist, even though some of these roles are still up for debate. Excessive activity in adults raises serum CK levels because it stresses muscle cells, according to studies [30]. Following an intramuscular injection, surgery, electromyography, or biopsy, it may also rise noticeably. Furthermore, CK levels in healthy people exhibit notable physiologic diversity depending on age, sex, and ethnicity, according to other studies [31]. CK increases drastically in skeletal muscle tissue experiencing high energy expenditure [25]. When exercising, it is known that CK levels increase significantly [17]. In addition, increased CK levels are also a sign of muscle damage.
Protein synthesis and degradation interact in a complicated and protracted way as a result of a series of events started by damaged muscle cells [32]. Therefore, muscle atrophy, muscle degeneration, and exercise-induced muscle damage (EIMD) can be caused by the breakdown of proteins [33]. Exercise-induced muscle injury results in altered protein structure, diminished muscular strength, and compromised muscle function. Increased serum levels of skeletal muscle enzymes (lactate dehydrogenase (LDH), myoglobin (Mb), and creatine kinase (CK), as well as decreased force output and neuromuscular deficiencies, are linked to exercise-induced muscle injury [34]. This muscle injury causes EIMD, which is manifested as limb edema, diminished neuromuscular function, and decreased range of motion [34]. These symptoms impair muscular function and make it difficult for athletes to engage in high-intensity training in the days that follow [35]. So maintaining training performance is very necessary, even additional nutrition is very important to maintain physical performance, so that it can be optimal in subsequent training.
Serum creatine kinase and lactate dehydrogenase are indicators of how well the skeletal muscles have adapted metabolically to physical exertion [36]. Serum levels of these enzymes that play a role in the metabolism of muscles, are typically quite low. These enzymes rise dramatically after severe exercise. After intense exercise, both athletes and healthy people have been shown to exhibit changes in blood muscle enzyme activity. Pyruvate is converted to lactic acid during exercise by the enzyme lactate dehydrogenase because active muscles utilize up available oxygen [37]. About 40% of the lactate in the blood is released by skeletal muscle, and the majority of this is subsequently taken up by the kidneys and liver, where it is converted to glucose by oxidation [38]. The muscle enzyme creatine kinase controls the conversion of creatine to phosphocreatine, which is then utilized to produce intracellular adenosine triphosphate [39]. However, during intense anaerobic exercise, creatine supplementation may boost ATP capacity and energy generation, potentially enhancing muscle strength, repetitions, and training volume. These factors may further enhance performance and muscle growth during the training period [40]. In addition, there is an interesting phenomenon of the relationship between CK and catalase (CAT), one of the endogenous antioxidants, during exercise.
CAT depletion happens first during exercise, reflecting rapid consumption to neutralize exercise-induced reactive oxygen species (ROS), whereas CK increases later, peaking after exercise as sarcolemmal damage from cumulative oxidative stress, mechanical tension, and ROS-induced inflammation allows the enzyme to leak into the bloodstream [41]. This temporal dissociation highlights the direct threat to membrane integrity posed by CAT's incapacity to fully counteract oxidative stress. This exacerbates CK release through mitochondrial dysfunction, lipid peroxidation, and protease activation, all of which contribute to the breakdown of muscle structure [42]. Consequently, CK is a delayed signal of permanent damage, but CAT is an early indicator of oxidative imbalance. This leads to a feedback loop in which unresolved oxidative stress (represented by prolonged suppression of CAT) exacerbates mechanical injury (represented by persistently high CK) [43]. As of right now, the rise in CK levels during exercise is a physiological reaction that aims to produce more ATP. To move other organs and perform skeletal muscle activity, the body needs to produce more ATP in order to achieve homeostasis.
Strenght and limitations
This systematic review's advantage is that it just examines randomized controlled trials, which eliminates the chance of unclear cause-and-effect linkages and being the most trustworthy form of scientific data. Furthermore, the samples collected are human-focused, exhibit consistent data, and aren't combined with samples from other categories, like animal samples.
We realize the limitations of this systematic review, we only focus on conducting an in-depth analysis of how exercise affects increased creatine kinase levels. In discussing physiological responses and their mechanisms, it may be felt to be less in-depth, but that is the limitation of researchers because they are aware of the complexity of physiological mechanisms that occur in the body. To examine it in other biomarkers such as how the stages of creatine kinase occur during physical exercise, experimental research is needed. In addition, a comparison between high and low intensity exercise also needs to be studied in depth, therefore further research is recommended to solve this problem.
CONCLUSIONS
- Regular exercise has been shown to dramatically raise creatine kinase levels, a biomarker of muscle injury, according to the associated papers we found.
- This is a normal physiological reaction to exercise. This happens as the body tries to produce more adenosin triphosphate in order to meet its needs.
Contributors:
Putro B.N. – data curation, visualization;
Wibawa J.C. – conceptualization, writing, original draft;
Ayubi N. – conceptualization, original draft.
Acknowledgement
We would like to express our gratitude that this publication has received funding from Universitas Sebelas Maret.
Funding. This research received no external funding.
Conflict of interests. The authors declare no conflict of interest.
REFERENCES
- Wibawa JC, Febrianto N, Fudin MS, Ockta Y, Festiawan R. The mechanism of physical exercise increases glutathione peroxidase as an endogenousantioxidant: a systematic review. Retos. 2025;63:610-9. doi: https://doi.org/10.47197/retos.v63.108856
- Wang F, Wang X, Liu Y, Zhang Z. Effects of Exercise-Induced ROS on the Pathophysiological Functions of Skeletal Muscle. Oxid Med Cell Longev. 2021 Oct 1;2021:3846122. doi: https://doi.org/10.1155/2021/3846122
- Wibawa JC, Setiawan A, Pratiwi DJ, et al. Increased activity of the catalase enzyme after physical exercise as a signal for reducing hydrogen peroxide (H2 O2): a systematic review. Fizjoterapia Pol. 2024;5:232-8. doi: https://doi.org/10.56984/8ZG020C7GDL
- Mendes S, Leal DV, Baker LA, Ferreira A, Smith AC, Viana JL. The Potential Modulatory Effects of Exercise on Skeletal Muscle Redox Status in Chronic Kidney Disease. Int J Mol Sci. 2023 Mar 23;24(7):6017. doi: https://doi.org/10.3390/ijms24076017
- Ayubi N, Wibawa JC, Sceisarriya VM, et al. Mechanism of physical exercise increases malondialdehyde levels as a marker of oxidative stress: A systematic review. Fizjoterapia2024;24(3):236-41. doi: https://doi.org/10.56984/8zg020a43v
- Chen J, Cheng F, Yang J, Yang J, Ran J, Liao Y. Effect of tissue flossing on eccentric exercise-induced muscle damage : A randomized controlled trial. Biol Sport. 2025 Feb 5;42(3):177-84. doi: https://doi.org/10.5114/biolsport.2025.147019
- Hotfiel T, Freiwald J, Hoppe MW, Lutter C, Forst R, Grim C, et al. Advances in Delayed-Onset Muscle Soreness (DOMS): Part I: Pathogenesis and Diagnostics. Sportverletzung-Sportschaden.2018 Dec;32(4):243-50. doi: https://doi.org/10.1055/a-0753-1884
- Owens DJ, Twist C, Cobley JN, Howatson G, Close GL. Exercise-induced muscle damage: What is it, what causes it and what are the nutritional solutions? Eur J Sport Sci.2019 Feb;19(1):71-85. doi: https://doi.org/10.1080/17461391.2018.1505957
- Schilder A, Hoheisel U, Magerl W, Benrath J, Klein T, Treede RD. Sensory findings after stimulation of the thoracolumbar fascia with hypertonic saline suggest its contribution to low back pain. Pain. 2014 Feb;155(2):222-31. doi: https://doi.org/10.1016/j.pain.2013.09.025
- Katayama H, Watanabe A, Machida T. A new perspective on tissue gliding dysfunction bordered by deep fascia as an indicator of delayed onset muscle soreness: A case report. J Bodyw Mov Ther. 2023 Oct;36:251-5. doi: https://doi.org/10.1016/j.jbmt.2023.06.014
- George MD, McGill NK, Baker JF. Creatine kinase in the U.S. population: Impact of demographics, comorbidities, and body composition on the normal range. Medicine(Baltimore). 2016 Aug;95(33):e4344. doi: https://doi.org/10.1097/MD.0000000000004344
- Brancaccio P, Maffulli N, Limongelli FM. Creatine kinase monitoring in sport medicine. Br Med Bull. 2007;81-82:209-30. doi: https://doi.org/10.1093/bmb/ldm014
- Baird MF, Graham SM, Baker JS, Bickerstaff GF. Creatine-kinase- and exercise-related muscle damage implications for muscle performance and recovery. J Nutr Metab.2012;2012:960363. doi: https://doi.org/10.1155/2012/960363
- Giechaskiel B. Weight Training and Creatine Kinase (CK) Levels: A Literature Review. Int J Sci Res. 2020;9(1):303-11. doi: https://doi.org/10.21275/ART20203985
- Koch AJ, Pereira R, Machado M. The creatine kinase response to resistance exercise. J Musculoskelet Neuronal Interact. 2014 Mar;14(1):68-77. PMID: 24583542
- Santos Cerqueira M, Kovacs D, Martins de França I, Pereira R, da Nobrega Neto SB, Aires Nonato RD, et al. Effects of Individualized Ischemic Preconditioning on Protection Against Eccentric Exercise–Induced Muscle Damage: A Randomized Controlled Trial. Sports Health.2021 Nov-Dec;13(6):554-64. doi: https://doi.org/10.1177/1941738121995414
- Allard NAE, Janssen L, Lagerwaard B, Nuijten MAH, Bongers CCWG, Rodenburg RJ, et al. Prolonged Moderate-Intensity Exercise Does Not Increase Muscle Injury Markers in Symptomatic or Asymptomatic Statin Users. J Am Coll Cardiol. 2023 Apr 11;81(14):1353-64. doi: https://doi.org/10.1016/j.jacc.2023.01.043
- Bray JW, Towlson C, Hayes SC, Fogarty M. Creatine kinase and neuromuscular fatigue responses following differing spells of simulated cricket fast bowling. PLoS One. 2025 Jan 22;20(1):e0317692. doi: https://doi.org/10.1371/journal.pone.0317692
- D'Alleva M, Sanz JM, Giovanelli N, Graniero F, Mari L, Spaggiari R, et al. The influence of prolonged aerobic exercise on cardiac, muscular, and renal biomarkers in trained individuals with obesity. Eur J Appl Physiol. 2025 May;125(5):1485-500. doi: https://doi.org/10.1007/s00421-024-05697-8
- Biss S, Teschler M, Heimer M, Thum T, Bär C, Mooren FC, et al. A single session of EMS training induces long-lasting changes in circulating muscle but not cardiovascular miRNA levels: a randomized crossover study. J Appl Physiol. 2023Apr1;134(4):799-809. doi: https://doi.org/10.1152/JAPPLPHYSIOL.00557.2022
- Beiter T, Erz G, Würden A, Nieß AM. Impact of moderate environmental heat stress during running exercise on circulating markers of gastrointestinal integrity in endurance athletes. Physiol Rep. 2025 Apr;13(7):e70305. doi: https://doi.org/10.14814/phy2.70305
- Kusmierczyk J, Wiecek M, Bawelski M, Szygula Z, Rafa-Zablocka K, Kantorowicz M, et al. Pre-exercise cryotherapy reduces myoglobin and creatine kinase levels after eccentric muscle stress in young women. Front2024 Jun 19;15:1413949. doi: https://doi.org/10.3389/fphys.2024.1413949
- Arriel RA, de Souza HLR, da Mota GR, Marocolo M. Declines in exercise performance are prevented 24 hours after post-exercise ischemic conditioning in amateur cyclists. PloS One. 2018 Nov 9;13(11):e0207053. doi: https://doi.org/10.1371/journal.pone.0207053
- Schlattner U, Tokarska-Schlattner M, Wallimann T. Mitochondrial creatine kinase in human health and disease. Biochim Biophys Acta. 2006 Feb;1762(2):164-80. doi: https://doi.org/10.1016/j.bbadis.2005.09.004
- Fechner A, Willenberg A, Ziegelasch N, Merkenschlager A, Kiess W, Vogel M. Creatine kinase serum levels in children revisited: New reference intervals from a large cohort of healthy children and adolescents. Clin Chim Acta. 2024 Jun 15;560:119726. doi: https://doi.org/10.1016/j.cca.2024.119726
- Brewster LM, Van Valkengoed I, van Montfrans GA. African Ancestry vs. Creatine Kinase to Predict Hypertension Control: Time for a Change? Am J Hypertens. 2021 Dec 1;34(12):1264-8. doi: https://doi.org/10.1093/ajh/hpab114
- Wallimann T, Wyss M, Brdiczka D, Nicolay K, Eppenberger HM. Intracellular compartmentation, structure and function of creatine kinase isoenzymes in tissues with high and fluctuating energy demands: The ‘phosphocreatine circuit’ for cellular energy homeostasis. Biochem J. 1992 Jan 1;281(Pt 1):21-40. doi: https://doi.org/10.1042/bj2810021
- Schlattner U, Tokarska-Schlattner M, Wallimann T. Metabolite Channeling: Creatine Kinase Microcompartments. 2ndElsevier Inc.; 2013. doi: https://doi.org/10.1016/B978-0-12-378630-2.00283-8
- Weiss RG, Gerstenblith G, Bottomley PA. ATP flux through creatine kinase in the normal, stressed, and failing human heart. Proc Natl Acad Sci U S A. 2005 Jan 18;102(3):808-13. doi: https://doi.org/10.1073/pnas.0408962102
- Bekkelund SI, Jorde R. Creatine kinase in relation to body fat in a Caucasian overweight and obese population. Scand J Clin Lab Invest. 2018 Feb-Apr;78(1-2):43-8. doi: https://doi.org/10.1080/00365513.2017.1408140
- Tahmasebi H, Asgari S, Hall A, Higgins V, Chowdhury A, Thompson R, et al. Influence of ethnicity on biochemical markers of health and disease in the CALIPER cohort of healthy children and adolescents. Clin Chem Lab Med. 2020 Mar 26;58(4):605-17. doi: https://doi.org/10.1515/cclm-2019-0876
- Shen L, Meng X, Zhang Z, Wang T. Physical exercise for muscle atrophy. Adv Exp Med Biol. 2018;1088:529-45. doi: https://doi.org/10.1007/978-981-13-1435-3_24
- Damas F, Nosaka K, Libardi CA, Chen TC, Ugrinowitsch C. Susceptibility to Exercise-Induced Muscle Damage: A Cluster Analysis with a Large Sample. Int J Sports Med. 2016 Jul;37(8):633-40. doi: https://doi.org/10.1055/s-0042-100281
- Therdyothin A, Phiphopthatsanee N. The Effect of Omega-3 on Mitigating Exercise-Induced Muscle Damage. Cureus. 2025 Apr 1;17(4):e81559. doi: https://doi.org/10.7759/cureus.81559
- Leite CDFC, Zovico VC, Rica RL, Barros BM, Machado AF, Evangelista AL, et al. Exercise-Induced Muscle Damage after a High-Intensity Interval Exercise Session: Systematic Review. Int J Environ Res Public Health. 2023 Nov 20;20(22):7082. doi: https://doi.org/10.3390/ijerph20227082
- Brancaccio P, Limongelli FM, Maffulli N. Monitoring of serum enzymes in sport. Br J Sports Med. 2006;40(2):96-7. doi:org/10.1136/bjsm.2005.020719
- Okon IA, Beshel JA, Owu DU, Orie NN, Jim AE, Edet LI. Moderate aerobic exercise improves haematological indices without altering cardio-metabolic enzyme activities in sedentary healthy young adults. BMC Sports Sci Med Rehabil. 2025 Feb 28;17(1):32. doi: https://doi.org/10.1186/s13102-025-01080-y
- Tappy L. Metabolism of sugars: A window to the regulation of glucose and lipid homeostasis by splanchnic organs. Clin Nutr. 2021 Apr;40(4):1691-8. doi: https://doi.org/10.1016/j.clnu.2020.12.022
- Antonio J, Candow DG, Forbes SC, Gualano B, Jagim AR, Kreider RB, et al. Common questions and misconceptions about creatine supplementation: what does the scientific evidence really show? J Int Soc Sports Nutr. 2021 Feb 8;18(1):13.
doi: https://doi.org/10.1186/s12970-021-00412-w - Kreider RB, Kalman DS, Antonio J, Ziegenfuss TN, Wildman R, Collins R, et al. International Society of Sports Nutrition position stand: Safety and efficacy of creatine supplementation in exercise, sport, and medicine. J Int Soc Sports Nutr. 2017 Jun 13;14:18. doi: https://doi.org/10.1186/s12970-017-0173-z
- Tu H, Li YL. Inflammation balance in skeletal muscle damage and repair. Front Immunol. 2023 Jan;14:1133355. doi: https://doi.org/10.3389/fimmu.2023.1133355.
- Zhang X, Liu Y, Li L, Ma W, Bai D, Dugarjaviin M. Physiological and metabolic responses of Mongolian horses to a 20 km endurance exercise and screening for new oxidative-imbalance biomarkers. 2025;15(9):1-18. doi: https://doi.org/10.3390/ani15091350
- Jena AB, Samal RR, Bhol NK, Duttaroy AK. Cellular Red-Ox system in health and disease: The latest update. Biomed Pharmacother. 2023 Jun;162:114606. doi: https://doi.org/10.1016/j.biopha.2023.114606


 UK
UK  EN
EN