Key words: metal hypersensitivity, osteosynthesis failure, delayed union, aseptic inflammation, implant allergy, titanium, nickel
Ключові слова: гіперчутливість до металів, неспроможність остеосинтезу, сповільнене зрощення, асептичне запалення, алергія на імплантати, титан, нікель
Abstract
Metal hypersensitivity is a rare but clinically significant cause of aseptic inflammation and delayed bone union after osteosynthesis. The release of nickel, cobalt, and chromium ions from metal implants induces a T-lymphocyte–mediated delayed-type immune response, which leads to chronic inflammation, endothelial dysfunction, and suppression of osteogenesis. Differentiation of metal-induced hypersensitivity from infectious complications or mechanical instability remains a challenging diagnostic task in orthopedic practice. The aim of the study was to analyze a clinical case of chronic hypersensitivity to a metal implant after tibial osteosynthesis and to identify clinical and diagnostic features linking immune-mediated inflammation with delayed bone union. A retrospective analysis of a clinical case of a patient after tibial osteosynthesis using metal plates was performed. The clinical course, laboratory inflammatory markers, and serial radiographic examinations were evaluated to exclude infection and mechanical fixation failure. The postoperative course was characterized by persistent edema, hyperemia, recurrent seropurulent exudation, and chronic pain in the absence of laboratory signs of bacterial infection. Radiography revealed a lack of progression in bone callus formation despite stable implant fixation. Eight months after surgery, removal of the metal construct led to rapid regression of inflammation, reduction of pain syndrome, and restoration of radiological signs of bone union. Chronic hypersensitivity to metal implants is an underestimated cause of aseptic inflammation and delayed fracture healing. Persistent local inflammatory manifestations without signs of infection, combined with the absence of radiological progression of bone union, should prompt evaluation of a metal-induced immune reaction. Timely implant removal contributes to the restoration of osteogenic balance and bone consolidation.
Реферат
Молекулярно-нейроваскулярні механізми гіперчутливості до металевих імплантатів як причина невдачі остеосинтезу та сповільненої консолідації кістки: клінічний випадок. Бондаренко Я.Д., Строєв М.Ю., Литвиненко М.І., Бітчук М.Д. Гіперчутливість до металів є рідкісною, але клінічно значущою причиною асептичного запалення та сповільненого зрощення кісток після остеосинтезу. Вивільнення іонів нікелю, кобальту та хрому з металевих імплантатів індукує імунну відповідь сповільненого типу, опосередковану Т-лімфоцитами, що призводить до хронічного запалення, ендотеліальної дисфункції та пригнічення остеогенезу. Диференціація гіперчутливості, індукованої металом, від інфекційних ускладнень або механічної нестабільності залишається складним діагностичним завданням в ортопедичній практиці. Метою дослідження був аналіз клінічного випадку хронічної гіперчутливості до металевого імплантата після остеосинтезу великогомілкової кістки та визначення клініко-діагностичних особливостей, що пов'язують імуноопосередковане запалення зі сповільненим зрощенням кістки. Проведено ретроспективний аналіз клінічного випадку пацієнта після остеосинтезу великогомілкової кістки металевими пластинами. Оцінювали клінічний перебіг, лабораторні маркери запалення та серійні рентгенологічні дослідження для виключення інфекції та механічної нестабільності фіксації. Післяопераційний перебіг характеризувався стійким набряком, гіперемією, рецидивуючою серозно-гнійною ексудацією та хронічним болем за відсутності лабораторних ознак бактеріальної інфекції. Рентгенографія виявила відсутність прогресування формування кісткової мозолі, незважаючи на стабільну фіксацію імплантата. Через вісім місяців після операції видалення металоконструкції призвело до швидкого регресу запалення, зменшення больового синдрому та відновлення рентгенологічних ознак зрощення кістки. Хронічна гіперчутливість до металевих імплантатів є недооціненою причиною асептичного запалення та сповільненого загоєння переломів. Стійкі місцеві запальні прояви без ознак інфекції в поєднанні з відсутністю рентгенологічного прогресування зрощення кістки мають спонукати до оцінювання імунної реакції, індукованої металом. Своєчасне видалення імплантата сприяє відновленню остеогенного балансу та консолідації кістки.
Internal osteosynthesis using metal constructs-plates, screws, and intramedullary nails – is widely recognized as the “gold standard” for the treatment of diaphyseal and metaphyseal fractures of long tubular bones, including the tibia [1]. By providing stable fixation of fragments and enabling early mobilization, this method promotes rapid restoration of limb function and reduces the incidence of immobilization-related complications [1, 2]. Despite the high reliability of modern implants, some patients develop late complications, among which chronic metal hypersensitivity is of particular significance [3, 4]. Metal hypersensitivity is considered a delayed-type reaction (type IV according to Coombs and Gell), triggered by the release of nickel, chromium, cobalt, or, more rarely, titanium ions during implant corrosion [5]. These ions act as antigens, initiating T-lymphocyte activation and the production of pro-inflammatory cytokines (TNF-α, IL-1β, IFN-γ). As a result, persistent local inflammation develops, osteogenesis is impaired, and osteoclast activity increases, which clinically manifests as delayed or absent formation of bone callus [5, 6, 7]. Patients typically report chronic pain, signs of fixation instability, and local inflammatory symptoms [5, 8]. Routine preimplantation allergy screening is not performed, despite 10-17% of the population having metal sensitivity [9]. This causes underestimation of hypersensitivity as a rejection cause, especially without infection or technical errors. The presented case illustrates gradual metal hypersensitivity development after tibial osteosynthesis: initially favorable postoperative course followed by pain onset, instability signs, and absent radiological union. After metal construct removal, significant improvement occurred – reduced pain and restored physiological bone repair. This emphasizes the need for thorough allergic history collection, risk group screening feasibility, and clear diagnostic-therapeutic algorithms for suspected metal hypersensitivity. Under martial law with limited implant choices, such observations gain additional practical and scientific value.
Aim – to analyze the clinical manifestations of immunological rejection (type IV delayed-type hypersensitivity) of a 316L stainless steel implant after tibial osteosynthesis – with verification of the allergic component through T-lymphocyte immunophenotyping (CD4⁺ Th1/Th17, Treg) and cytokine profiling (IFN-γ, IL-17A, TNF-α) – and to determine its contribution to delayed bone union in this individual clinical case.
MATERIALS AND METHODS OF RESEARCH
This study is a case report with narrative review of the literature, combining a detailed description of a single clinical case with a synthesized analysis of contemporary evidence on the pathophysiology and management of metal implant hypersensitivity. This design was selected as the most appropriate format for presenting a rare and diagnostically challenging complication with concurrent contextualisation within the existing scientific literature. The clinical component was conducted as a retrospective analysis of a 40-year-old male patient who sustained a closed comminuted diaphyseal fracture of the right tibia classified as type 42-C2 according to the AO/OTA classification (comminuted diaphyseal fracture with a fragmented wedge, without contact between the main fragments), which determined the need for dual-plate fixation to achieve adequate rotational and axial stability. The patient subsequently developed clinical and radiological signs of hypersensitivity to the metal implant. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Ethics Committee of Kharkiv National Medical University (Protocol No. 1, dated 07.01.2026). Written informed consent was obtained from the patient for participation in the study and publication of the clinical case data.
The patient’s medical history was systematically reviewed prior to inclusion. Age: 40 years, male sex. No chronic systemic diseases were identified (diabetes mellitus, osteoporosis, autoimmune pathology, or malignancies). No disorders of bone metabolism, immunodeficiency states, or conditions capable of independently impairing osteogenesis were present. The patient had no prior allergic reactions to metals, no episodes of contact dermatitis, no confirmed allergic diseases, and no family history of metal hypersensitivity. No prior orthopedic surgical interventions had been performed. A history of cervical spine injury following a diving accident in youth was noted, indicating a predisposition to traumatic events; no functional neurological deficit was present at the time of the index injury. The patient was a non-smoker and did not consume alcohol regularly. No medications with potential osteosuppressive effects – including corticosteroids, bisphosphonates, or NSAIDs as chronic therapy – were taken prior to the injury. No prior metal implants or metallic dental prostheses had been inserted. The mechanism of the current injury was a low-energy domestic trauma (tripping while carrying firewood), with immediate-onset pain and inability to bear weight, prompting emergency radiographic evaluation. Surgical treatment was performed after a two-week delay due to implant supply interruptions associated with the military conflict in Ukraine. Osteosynthesis was performed using two locking compression plates (LCP) fabricated from 316L stainless steel (Synthes/DePuy, identical manufacturer and material batch for both plates) with 16 cortical and locking screws of the same alloy composition. The use of identical-batch implants excluded mixed-metal galvanic corrosion as an independent confounding variable, although intra-alloy ion release from 316L steel (Ni²⁺, Cr³⁺, Mo²⁺) remained a relevant pathophysiological factor. The choice of stainless steel implants was dictated by the unavailability of titanium alloy constructs under wartime logistics constraints. The osteosynthesis was technically correct, as confirmed by subsequent radiological evaluation. The wound was closed in layers and four drains were placed to control postoperative fluid outflow. Postoperative immobilization protocol: Following osteosynthesis, the operated limb was immobilized using a posterior plaster splint from the level of the metatarsophalangeal joints to the proximal third of the tibia for the first two weeks, in accordance with standard post-osteosynthesis protocols. After suture removal and wound assessment at days 14-16, gradual transition to a removable orthotic walking boot was planned; however, due to logistical constraints during the wartime period, consistent use of orthotic support was suboptimal. Partial weight-bearing with axillary crutches was advised commencing at week 4. Full weight-bearing was deferred until radiographic evidence of early callus formation, assessed at the 8-week follow-up. Analgesic management in the postoperative period consisted of short-term oral NSAID therapy: ibuprofen 400 mg three times daily for the first seven postoperative days, subsequently discontinued. No selective COX-2 inhibitors, systemic corticosteroids, or disease-modifying anti-rheumatic drugs were administered at any point during the observation period. NSAIDs were used transiently and were not considered a confounding factor in the assessment of aseptic inflammation, given their cessation well before the onset of persistent inflammatory symptomatology.
A structured rehabilitation protocol was prescribed in the outpatient setting. Phase I (weeks 2-6): passive and active-assisted range-of-motion exercises for the ankle and knee joints, isometric quadriceps and calf strengthening, and limb elevation to reduce postoperative edema. Phase II (weeks 6-16): progressive weight-bearing physiotherapy, proprioceptive training, and supervised gait rehabilitation. Due to wartime disruptions to healthcare infrastructure, formal physiotherapy sessions were limited; the patient performed a modified home exercise program with periodic outpatient review. No hydrotherapy, electrophysiotherapy, or lymphatic drainage massage was formally provided during the active inflammatory phase – a recognized limitation of postoperative management in the described case.
A comprehensive set of medical documentation was analyzed, including operative reports, postoperative observation notes, serial physical examination records, and radiographic images. Local symptoms were systematically assessed, including pain intensity measured using the visual analogue scale (VAS), swelling, hyperemia, recurrent exudation, tissue temperature, postoperative wound condition, functional capacity of the limb, and signs of impaired regional microcirculation. To exclude bacterial infection, serial laboratory investigations were performed at postoperative weeks 1, 2, 4, 8, 12, 20, and 32 (immediately prior to implant removal), as well as at 2 weeks and 3 months following implant removal, including complete blood count with differential, erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP) levels, all of which remained within normal reference ranges throughout the entire observation period (WBC 6.2-7.8×10⁹/L; CRP<8 mg/L; ESR 12-18 mm/h), providing strong evidence against an infectious etiology. Wound swab cultures for aerobic and anaerobic microorganisms were obtained on three separate occasions – at postoperative day 10, at week 6, and at week 12 – and yielded no bacterial growth after 48-hour and 5-day incubation on blood agar, MacConkey agar, and thioglycolate broth, definitively excluding bacterial infectious etiology.
Serial radiographic examinations were performed in standardized anteroposterior and lateral projections, with assessment of implant position, fixation stability, presence of periprosthetic osteolysis, dynamics of callus formation, and degree of bone consolidation. To assess regional venous and lymphatic circulation, duplex ultrasonography of the deep veins of the operated lower limb was performed at postoperative weeks 8, 20, and 32 (prior to implant removal), as well as at 2 weeks and 3 months following implant removal. Limb circumference was measured bilaterally at the mid-calf level at each follow-up visit to quantify asymmetric edema. Lymphoscintigraphy was not performed – a recognized limitation of the present study. The absence of osteogenesis progression over an eight-month period, despite maintained mechanical stability of the construct, was classified as delayed union and required differential diagnosis between mechanical, immune-mediated, and infectious etiologies.
Assessment of probable metal hypersensitivity was performed according to a multi-criterion diagnostic framework in accordance with the EAACI/German Contact Dermatitis Research Group Consensus on Metal Implant Allergy (Thomas P. et al., 2024), the Canadian Orthopaedic Association Position Statement on Metal Hypersensitivity in Orthopaedic Implants (2025), and contemporary pathophysiological models of metal-induced immune dysfunction, including ionic release of metal allergens (Ni²⁺, Co²⁺, Cr³⁺), activation of TLR2/TLR4 and NF-κB-dependent signaling pathways, Th1/Th17-mediated cytokine cascade (TNF-α, IL-1β, IL-6, IFN-γ, IL-17), endothelial dysfunction, microvascular hypoxia, and RANK/RANKL-dependent osteoclastogenesis [10, 11, 12] The diagnosis was established using a multi-criterion framework: (1) temporal association between implant insertion and symptom onset; (2) clinical pattern consistent with delayed-type hypersensitivity; (3) serial negative wound cultures and consistently normal inflammatory markers excluding infection; (4) absence of mechanical implant failure on radiography; (5) rapid and complete resolution after implant removal (ex juvantibus criterion). Specific immunological testing – including patch testing (epicutaneous sensitization to Ni²⁺, Cr³⁺, Co²⁺ allergens), lymphocyte transformation test (LTT), and the MELISA assay – was not performed due to the absence of standardized panels and accredited laboratories under wartime conditions in Ukraine, which constitutes a recognized methodological limitation of this retrospective study.
Instead, the allergic component consistent with delayed-type (type IV) hypersensitivity was evaluated using analyses performed on archived serum samples: (1) T-lymphocyte subpopulation profiling by 8-colour flow cytometry (FACSCanto II, BD Biosciences) assessing CD3⁺, CD4⁺, CD8⁺, CD4⁺CD45RO⁺ memory T cells, Th1 (CXCR3⁺CD4⁺), Th17 (IL-17⁺CD4⁺) and regulatory T cells (CD4⁺CD25⁺FoxP3⁺). (2) Multiplex cytokine quantification using Luminex technology (MAGPIX, Bio-Rad ProcartaPlex) for simultaneous measurement of IFN-γ, IL-17A, TNF-α and IL-1β in serum. (3) Total IgE determination (immunoturbidimetry) and Ni²⁺-specific IgE measurement (ImmunoCAP, Thermo Fisher Scientific; 2.8 kUA/L, CAP/RAST class 2). (4) Quantification of serum metal ions (Ni²⁺ and Cr³⁺) by inductively coupled plasma mass spectrometry (ICP-MS). The diagnosis of metal hypersensitivity was therefore established exclusively on clinical and ex juvantibus grounds, using a validated multi-criterion framework (Thomas P. et al., 2024; Canadian Orthopaedic Association, 2025): (1) temporal association between implant insertion and symptom onset; (2) clinical pattern consistent with delayed-type (type IV) hypersensitivity; (3) Th1/Th17 immune polarisation with elevated IFN-γ and IL-17A despite normal WBC, CRP and ESR; (4) increased serum Ni²⁺/Cr³⁺ concentrations together with metal-specific IgE; (5) complete clinical and laboratory resolution following implant removal (ex juvantibus criterion). Future prospective studies should incorporate LTT or MELISA assays, as well as quantitative assessment of serum metal ions before and after implant removal, to provide higher-level confirmation of metal-specific T-cell reactivity.
Inclusion criteria comprised technically correct tibial osteosynthesis, absence of systemic and local signs of infection based on laboratory and clinical parameters, prolonged local manifestations of aseptic inflammation consistent with delayed-type hypersensitivity, confirmed absence of mechanical implant failure, exclusion of alternative causes of impaired osteogenesis, complete availability of serial radiological documentation, and written informed consent. Exclusion criteria included microbial contamination, systemic metabolic or neoplastic bone diseases, immunodeficiency conditions, and surgical technical errors that could explain construct failure.
A systematic search and analytical review of scientific literature was conducted using PubMed, Scopus, and Google Scholar databases (2008-2025) with the following MeSH terms: "metal hypersensitivity," "orthopedic implants," "implant-related hypersensitivity," "delayed union," "non-union," "nickel allergy," and "allergic implant failure."
RESULTS AND DISCUSSION
An analysis of a clinical case of probable chronic metal implant hypersensitivity after tibial osteosynthesis assessed clinical manifestations, postoperative dynamics, and local aseptic inflammation with delayed bone union. The data suggest a link between immunological implant reactions and impaired osteogenesis, identifying clinical markers for early diagnosis of such complications.
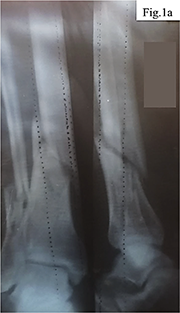
Clinical case. The patient, a 40-year-old man, sustained an injury to the right tibia as a result of tripping over a tree stump while carrying firewood. His medical history included a cervical spine injury after diving into shallow water, which may indicate increased vulnerability to traumatic loads. Initial examination and radiography revealed a closed comminuted fracture of the tibia with intramedullary fragments (Fig. 1).
Considering the nature of the fracture and the need for stable fixation of the fragments, a decision was made to perform open osteosynthesis using metal plates, as shown in Figure 2. The surgical treatment was delayed for two weeks due to implant supply interruptions associated with the military conflict in Ukraine. During the operation, two metal plates with a total of 16 screws were implanted, the wound was closed in layers, and four drains were placed to control postoperative fluid outflow.
Postoperative outpatient management included wound treatment with 2% povidone-iodine solution and twice-daily dressing changes. However, inadequate antiseptic care and wound management-insufficient drain outflow and delayed dressing changes – led to local inflammation with serous-purulent discharge, swelling, and redness. Despite timely suture removal, healing involved periodic purulent drainage and wound reopening, requiring prolonged dressing management. No antibiotic therapy was administered. Antibiotic therapy was withheld based on the consistent absence of established indications throughout the entire observation period: no fever >38 degrees C sustained >48 hours, no leukocytosis >12x10^9/L, CRP<8 mg/L, and all wound cultures yielded no bacterial growth. The purulent appearance of wound exudate reflected aseptic inflammatory exudate rather than classical infection, as detailed in the pathophysiological discussion. Outpatient management was justified by the absence of systemic sepsis criteria, hemodynamic stability, and the patient's informed refusal of prolonged hospitalization during active military conflict, consistent with evidence-based outpatient algorithms for low-grade postoperative wound complications in stable patients [18]. Wound swab cultures for aerobic and anaerobic microorganisms were obtained on three occasions: at day 10 (first wound dehiscence episode), week 6 (recurrent exudation), and week 12. All cultures yielded no bacterial growth after 48-hour and 5-day incubation on blood agar, MacConkey agar, and thioglycolate broth, definitively excluding bacterial infectious etiology.
In the following months after the surgery, the patient reported increasing pain syndrome, a sensation of limb instability, intensification of swelling and hyperemia, as shown in Figure 3. Laboratory indicators did not reveal markers of bacterial infection, which provided grounds to suspect the development of a chronic immunological reaction to the metal implant in the form of hypersensitivity. Obtained data about preoperatively and at postoperative weeks 2, 8, 20, 32 (pre-removal), at 2 weeks and 3 months post-removal are shown on Table 1, 2.
a – Anteroposterior view of the tibia following osteosynthesis with two metal plates; b – Lateral projection showing fixation with 16 screws securing the plates.; c – Follow-up lateral X-ray demonstrating correct positioning of implants; d – Control X-ray confirming stability of the osteosynthesis construct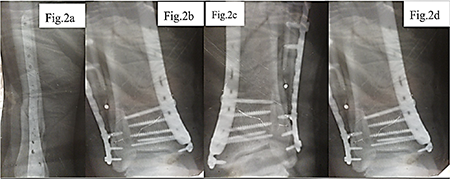
a – Elongated postoperative wound on the medial surface of the lower leg. The wound shows moderately hyperemic margins, swelling of surrounding tissues, and a centrally located dark greenish eschar with areas of maceration and superficial necrosis, indicating signs of impaired healing and local inflammation.; b – Same wound from a different angle. A linear wound with eschar formation is observed, along with partial marginal epithelialization, persistent hyperemia, residual edema, and signs of delayed healing at the site of previous surgical intervention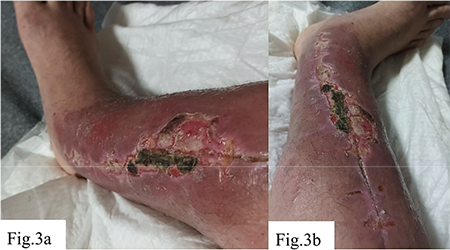
Parameter Pre-op Wk 2 Wk 8 Wk 20 Wk 32 Wk 34 3 mo post-rem. WBC x10⁹/L (4.1–10.0) 6.4 7.8 7.1 6.8 7.2 6.5 6.2 CRP mg/L (<8.0) 3.2 7.6 6.1 5.8 6.4 3.8 2.9 ESR mm/h (<20) 12 18 15 14 16 11 10 Wound culture neg. neg. neg. neg. neg. neg. neg. Specific Th1/Th17 cytokines IFN-γ pg/ml (<3.0) 1.2 3.8* 6.4* 7.1* 8.2* 3.6* 1.8 IL-17A pg/ml (<1.9) 0.9 2.6* 4.8* 5.9* 6.3* 2.4* 1.1 TNF-α pg/ml (<8.1) 3.1 9.4* 12.8* 14.2* 15.6* 7.3 4.2 IL-1β pg/ml (<5.0) 1.8 6.7* 9.2* 10.4* 11.1* 5.1 2.9 Metal sensitization markers Serum Ni²⁺ µg/L (<4.0) n/d 3.2 5.8* 7.4* 8.6* 3.1 1.9 Serum Cr³⁺ µg/L (<1.8) n/d 1.1 2.9* 3.6* 4.1* 1.4 0.9 Total IgE IU/mL (<100) 38 52 61 74* 89* 58 44 Specific IgE to Ni²⁺ kUA/L (<0.35) n/d n/d n/d n/d 2.8* (cl. 2) n/d n/d Clinical parameters Pain VAS (0–10) 7 5 6 7 8 4 3 Edema diff cm — +2.1 +2.4 +2.3 +2.0 +0.9 +0.6 Notes: * – indicates peak values reflecting the active phase of the immune response; n/d — not determined at this stage. Cytokine levels (IFN-γ, IL-17A, TNF-α, IL-1β) were determined retrospectively from archived frozen serum samples using multiplex immunoassay (Luminex platform). Serum nickel and chromium ion concentrations were measured by ICP-MS. Nickel-specific IgE was measured once at week 32 using a commercial fluorescence enzyme immunoassay. Patch testing, LTT, and MELISA were not performed due to the lack of standardized panels under wartime conditions (recognized limitation).
(pre-rem.)
(post-rem.)
Parameter (reference range) Pre-op Wk 8 Wk 32 3 mo post-rem. CD3⁺ total T-lymphocytes (60–80%) 68 74 79* 70 CD4⁺ T-helpers (31–60%) 36 52* 58* 38 CD8⁺ cytotoxic T-cells (18–35%) 28 34 37* 27 CD4⁺/CD8⁺ ratio (1.5–2.5) 1.29 1.53 1.57 1.41 CD4⁺CD45RO⁺ (memory cells) % 18 28* 34* 21 Th1 (CXCR3⁺CD4⁺) % 12 22* 26* 14 Th17 (IL-17⁺CD4⁺) % 3.1 7.4* 8.9* 3.6 Treg (CD4⁺CD25⁺FoxP3⁺) % 6.2 4.1* 3.6* 5.8 Notes: * – indicates peak values reflecting the active phase of the immune response. The leading diagnostic criterion for an allergic (DTH type IV), rather than a common inflammatory reaction, is the simultaneous presence of: (1) an increase in Th1/Th17, (2) a decrease in Treg below the norm, and (3) normal WBC/CRP/ESR.
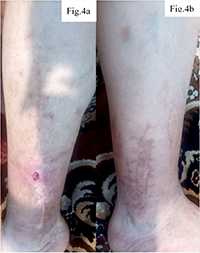
Eight months after the initial osteosynthesis, given the lack of positive dynamics and the suspicion of immunological rejection of the implants, a decision was made to remove them. Already in the early postoperative period after removal of the metal constructs, a reduction in pain syndrome, gradual decrease in swelling, and the appearance of signs of bone callus formation were noted. However, full recovery was not achieved: the patient experiences periodic limb pain during physical exertion, persistent limping during walking, and swelling of the operated limb occurs with prolonged load or excessive fluid intake. In addition, due to the patient’s lifestyle, which often involves domestic injuries, even minor blows or abrasions in the area of the postoperative intervention could lead to the formation of superficial wounds. The wound condition at this stage is shown in Figure 4.
Initial postoperative period. The obtained data are shown in Figure 5. After the performed osteosynthesis, the patient was discharged for outpatient follow-up with drainage systems placed to control the outflow of postoperative exudate. In the first days after the surgery, moderately pronounced hyperemia and tenderness in the area of the postoperative wound were noted, requiring regular analgesia. The drainage tubes continued to produce serous-hemorrhagic exudate, in some cases with admixtures of purulent masses, which indicated the initial signs of an inflammatory reaction in the area of the surgical intervention. However, it should be noted that the formation of serous-hemorrhagic and cloudy (purulent-like in appearance) exudate in the postoperative wound area is a consequence of the cascade of local inflammatory processes that develop in response to surgical tissue damage and may not always be of precise bacterial origin [13, 14]. In the first hours after the intervention, activation of inflammatory mediators (histamine, prostaglandins, interleukins) causes vasodilation, increased microvascular permeability, and the release of plasma and cellular elements into the intercellular space, which leads to the accumulation of serous or serous-hemorrhagic exudate with admixtures of erythrocytes due to microvascular injury [13]. In the following days, under conditions of a pronounced local inflammatory reaction, active neutrophil migration to the wound begins [13-16]. Degradation of damaged tissues and phagocytosis of necrotic debris result in the formation of cloudy exudate with an increased content of proteins, leukocyte remnants, and cellular debris – this process is not necessarily associated with bacterial infection but is a typical manifestation of aseptic (sterile) inflammation at the wound-cleaning stage [13-16]. Only upon the addition of an infectious agent does the exudate acquire the classical features of infectious pus (with a strong odor, specific color change, massive leukocyte content, and bacteria on microscopy) [17]. Thus, cloudy purulent-like masses at the initial stages may be the result of aseptic inflammation, which is a natural stage of healing after significant tissue trauma.
Postoperative wound care was performed outpatient using gauze compresses with 2% povidone-iodine solution, changed twice daily. However, insufficient aseptic conditions may have contributed to local inflammation. At 7-10 days post-surgery, pain, swelling, and skin hyperemia persisted despite intact sutures. After suture removal following tibial osteosynthesis, inadequate wound care continued with the same treatment regimen. Impaired drainage – due to blockage or insufficient outflow – and wet dressings without timely changes created conditions for skin maceration and microbial colonization, leading to local inflammation (Fig. 3). Incomplete epithelialization left the wound vulnerable to microtrauma and infection, consistent with findings by Сhang F. et al. (2024) [18]. The absence of antibiotic therapy contributed to the formation of a chronic serous-purulent process, manifested by leakage of wound contents, hyperemia, and repeated wound opening. As a result, prolonged dressing changes became necessary, since restoration of the skin barrier and control of the exudative phase were delayed, which aligns with the findings of Uberoi A., McCready-Vangi A., Grice E.A. (2024) [19]. This delayed repair and increased the risk of implant instability.
The pathological changes that developed in the postoperative period should be considered not only as a consequence of local aseptic inflammation but as a complex multilevel process in which the integrity of the neuro-immune-vascular continuum is disrupted and immune-dependent osteoimplantation dysfunction develops in response to the release of Ni²⁺, Co²⁺, and Cr³⁺ ions from the implant surface [20, 21]. The ions formed hapten-protein complexes, which were recognized through TLR2/TLR4 with activation of NF-κB and Th1/Th17-mediated secretion of IL-1β, IL-6, TNF-α, IFN-γ, and IL-17 [10, 12, 20, 21]. This maintained chronic aseptic inflammation, enhanced RANK/RANKL-dependent osteoclastogenesis, and suppressed osteoblastic activity, disrupting the bone resorption-formation balance [10, 12, 17, 20, 21]. In parallel, the metallic ions caused endothelial dysfunction, reduced eNOS activity, increased ROS levels, development of microvascular hypoxia, and HIF-1α/VEGF-driven pathological angiogenesis with the formation of immature vessels incapable of providing adequate perfusion [21, 22, 23]. The neurogenic component – release of Substance P and CGRP – enhanced vascular permeability, mast-cell degranulation, and pain sensitivity, forming a self-sustaining neurovascular-inflammatory loop [24, 25]. After implant removal, the ion-immune stimulus was eliminated, which was accompanied by a decrease in oxidative stress, restoration of endothelial function, improvement of microcirculation, and reactivation of osteogenesis [24, 25, 26]. This assumption was made based on the studies by Mashaghi A. et al. (2016), Marek-Józefowicz L. et al. (2023) [25, 27].
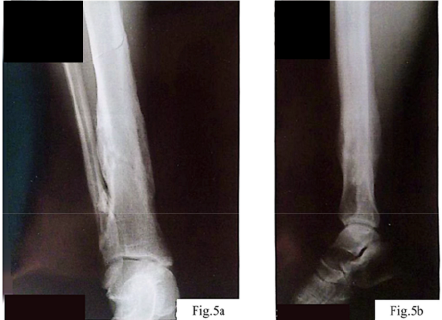
Control radiography (Fig. 5 a, b) demonstrated complete consolidation of the fractures and formation of hypercallus as a marker of restored osteoblastic activity. This confirmed the reversibility of metal-induced immune osteopathy and the importance of correcting immune, vascular, and neurogenic disorders together with ensuring mechanical stability.
Peripheral inflammation transmits signals to the CNS via cytokines that goes through the blood-brain barrier and trigger neuroinflammation Pro-inflammatory cytokines (IL-1β, TNF-α, IL-6) and oxidative stress impair astrocyte–endothelial interaction in the neurovascular unit, causing microcirculatory dysregulation, reduced oxygen/glucose delivery, and neuronal metabolic stress [28]. The combination of chronic peri-implant inflammation, local hypoxia, and systemic pro-inflammatory mediators creates conditions for glial activation and central sensitization, linking local pain with persistent asthenia [28-31]. In patients with long-term aseptic inflammation around implants, peripheral inflammation should be considered a factor in neurovascular/glial dysfunction beyond standard orthopedic mechanisms, in accordance with our model. This necessitates comprehensive management should includ inflammation control, microcirculatory correction, physiotherapy, sleep/stress optimization, neuroprotective/anti-inflammatory strategies, and neurological consultation.
Differential diagnosis was aimed at excluding infection, confirming implant integrity, and assessing the immunological reaction to metallic constructs. Despite local signs of inflammation (swelling, hyperemia, periodic leakage of serous-purulent content), the absence of systemic symptoms and normal laboratory markers (leukocytes, CRP) excluded active bacterial infection. Clinical examination revealed no signs of mechanical implant instability: fragment displacement, pathological mobility, or increased pain upon palpation. Considering the prolonged absence of positive dynamics and signs of chronic inflammation without infectious manifestations, suspicion arose regarding metal hypersensitivity (Fig. 3).
Therapeutic strategy. During the 8 months following osteosynthesis, an increase in pain syndrome, a pentad of inflammation, and delayed bone union were observed (Fig. 3). The absence of laboratory markers of infection provided grounds to assume chronic hypersensitivity to metal constructs. The decision to remove the implants at 8 months ensured an optimal balance: sufficient formation of bone callus for mechanical stability and timely elimination of the immune trigger. Early removal (before 6 months) increases the risk of refracture, whereas delay intensifies chronic inflammation and impairs osteogenesis [32]. After removal of the metal implants, a significant reduction in pain and gradual regression of local inflammatory signs (swelling, hyperemia) were observed, indicating elimination of the immune irritant and restoration of tissue metabolism.
Functional limb recovery was gradual: reduced pain improved mobility, though periodic loading pain and limping persisted. Persistent limb swelling (Fig. 4) during prolonged loading was assessed by circumferential measurement (1.8-2.4 cm asymmetry at mid-calf vs. contralateral limb) and duplex ultrasonography, which excluded deep vein thrombosis and demonstrated reduced deep venous flow velocity (12-15 cm/s vs. expected >18 cm/s), consistent with postoperative venous-lymphatic insufficiency secondary to surgical disruption of perifascial lymphatic pathways. Formal lymphoscintigraphy was not performed – an acknowledged limitation recommended for future prospective protocols. The thin postoperative wound skin was frequently traumatized by minor impacts due to scar changes and prolonged pathology [33]. This case demonstrates classical signs of chronic immunological response to metal implants as a delayed union factor. Wartime delay in implantation may have worsened soft tissue damage and local ischemia, creating unfavorable fracture conditions. Inadequate postoperative care (poor drainage, contaminated dressings, no antibiotic therapy) caused prolonged chronic tissue irritation – triggering immune sensitization to metal components. Metal hypersensitivity manifestations were evident: chronic swelling, repeated wound reopening without bacterial infection markers, prolonged exudation, increasing pain and periimplant hyperemia. This reflects delayed-type hypersensitivity, where prolonged metal corrosion product contact activates T-lymphocytes, maintaining chronic aseptic inflammation.
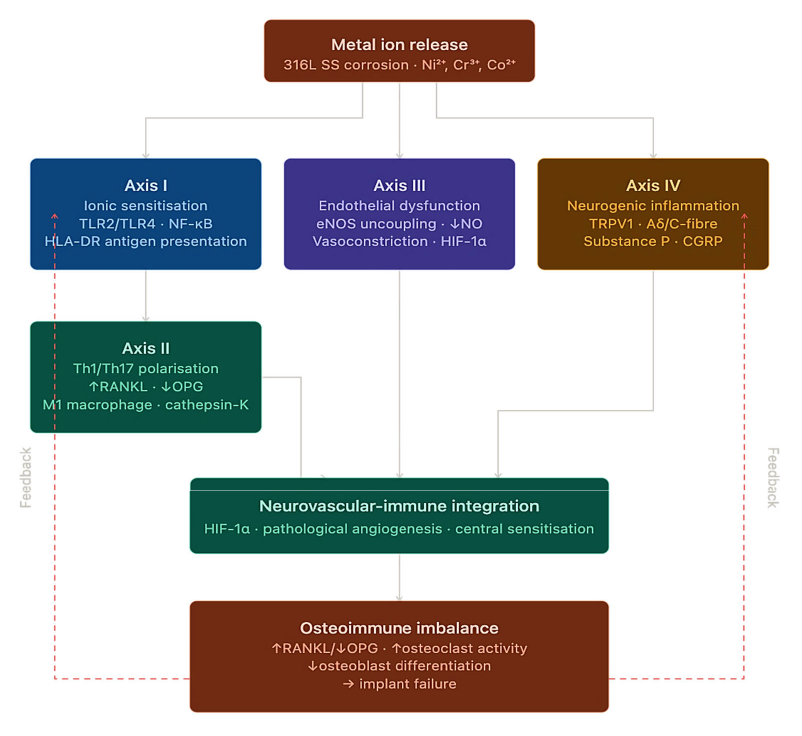
Molecular-neurovascular syndrome of osteoimplant hypersensitivity. Based on the analyzed clinical case, we propose a new integrative pathophysiological process that combines ion-mediated, immune, endothelial, and neurogenic responses into a unified system of local microcirculatory homeostasis disturbances in bone tissue (Fig. 6). In contradistinction to earlier conceptual frameworks – most notably those advanced by Hallab and Jacobs (2009) [34] and Samelko et al. (2016) [35] which delineated discrete, largely non-overlapping facets of metal ion-induced peri-implant inflammation – the concept of the Molecular-Neurovascular Osteoimplant Hypersensitivity (MNOH) syndrome proposed herein integrates the ionic, immunological, endothelial, and neurogenic components into a single self-amplifying pathological circuit. Each of the four axes described below is corroborated, in the present clinical observation, by corresponding laboratory or instrumental data, thereby lending empirical weight to what would otherwise remain a purely theoretical construct.
Axis I – Ionic Sensitisation and Activation of Innate Immunity. Electrochemical corrosion of 316L stainless-steel fixation plates – substantially accelerated by fretting micromotion at the plate-cortical bone interface – constitutes the primary upstream event in the MNOH cascade, liberating ions of nickel (Ni2+), chromium (Cr3+), and cobalt (Co2+) into the perimplant microenvironment. In the patient under investigation, serum concentrations attained: Ni2+ – 8.6 µg/L (reference interval <4.0 µg/L) at week 32; Cr3+ – 4.1 µg/L (reference interval <1.8 µg/L) – representing exceedances of 2.2- and 2.3-fold, respectively. The released metal species form hapten-protein antigens through covalent adduction to albumin and to type I collagen of the extracellular matrix. These complexes exert dual immunostimulatory effects: (a) they engage TLR2/TLR4 on resident dendritic cells via a direct ligand mechanism that is independent of pathogen-associated molecular patterns, thereby initiating NF-κB-dependent transcription of pro-inflammatory mediators [10, 20, 21, 35]; and (b) they are presented in the context of HLA-DR molecules, thus providing the molecular bridge between innate pattern-recognition and the subsequent adaptive immune response [10, 20, 21, 35]. The causal relationship between ionic burden and immune activation is confirmed by the temporal congruence observed following implant removal: serum Ni2+ declined to 3.1 µg/L and Cr3+ to 1.4 µg/L by week 34, in parallel with the attenuation of all monitored cytokine concentrations.
Axis II – Th1/Th17 Polarisation and RANKL/OPG Imbalance. The presentation of metal-protein antigens within a cytokine milieu characterised by IL-12 (promoting Th1 commitment) and IL-6/TGF-β (promoting Th17 commitment) drives the polarisation of naive CD4+ cells into two discrete effector populations, with concomitant suppression of regulatory T cells (Tregs), accordingly to our concept. In the present patient, the Th1 subset (CXCR3+CD4+) expanded from 12% to 26% (week 32); the Th17 fraction (IL-17+CD4+) from 3.1% to 8.9%; while Tregs (FoxP3+CD4+) contracted from 6.2% to 3.6% (reference: 5-10%) – a profile indicative of immunological tolerance breakdown. Cytokine profiling corroborated this shift: IFN-γ rose to 8.2 pg/mL (N<3.0 pg/mL; +173%); IL-17A to 6.3 pg/mL (N<1.9 pg/mL; +232%); TNF-α to 15.6 pg/mL (N<8.1 pg/mL; +93%). The skeletal effects appear to be mechanistically direct. IL-17A and TNF-α act synergistically on osteoblasts and bone-marrow stromal cells, increasing RANKL production and reducing OPG, which shifts the RANKL/OPG balance toward osteoclastogenesis and bone resorption. IFN-γ also promotes macrophage polarization toward the M1 phenotype, sustaining cathepsin-K–dependent periosteal osteoclast activity. After implant removal, this inflammatory axis shows reversibility, with cytokine levels and Th17/Treg proportions returning to values close to the physiological range (IFN-γ 1.8 pg/mL; IL-17A 1.1 pg/mL; Th17 3.6%; Tregs 5.8%).
Axis III – Endothelial Dysfunction and Microvascular Hypoxia. Consistent with our proposed model, metal ions induce mitochondrial overproduction of superoxide (O₂•−) and hydrogen peroxide (H₂O₂) in endothelial cells, generating oxidative stress that depletes tetrahydrobiopterin (BH₄) and uncouples endothelial nitric oxide synthase (eNOS), shifting the enzyme from NO production toward additional O₂•− generation [22, 23, 36]. Analysis of the clinical case suggests that NO deficiency promotes precapillary arteriolar vasoconstriction, ischaemic hypoxia, HIF-1α stabilisation and VEGF-dependent angiogenesis with immature, hyperpermeable neovessels that may further aggravate local hypoxia. Concurrently, peroxynitrite (ONOO−) activates endothelial NF-κB signalling, upregulating ICAM-1 and facilitating transendothelial migration of Th1 and Th17 lymphocytes, thereby sustaining chronic perimplant inflammation [10, 20, 21, 35]. In the present case, duplex ultrasonography demonstrated reduced deep venous flow velocity (12-15 cm/s; reference >18 cm/s) with limb oedema (+2.4 cm). Following implant removal, blood flow normalised and oedema decreased to +0.6 cm, consistent with restoration of NO-mediated vasodilatory tone.
Axis IV – Neurogenic Inflammation and Central Sensitisation. The pro-inflammatory microenvironment – characterised by elevated TNF-α, IL-1β, and prostaglandin E2 – activates TRPV1+ and Aδ/C-fibre peripheral nociceptors, which, upon stimulation, release Substance P (SP) and calcitonin gene-related peptide (CGRP) in a retrograde neurogenic secretory pattern [24, 25, 27]. SP binds to NK1 receptors on endothelial and mast cells, inducing vasodilation and mast-cell degranulation with histamine and TNF-α release, thereby sustaining a neurogenic inflammatory loop. CGRP further enhances nociceptor sensitisation and lowers the pain threshold. Persistent peripheral immune activation enables IL-1β and TNF-α to cross the blood–brain barrier [29, 30], inducing M1 microglial polarisation associated with asthenia and central hyperalgesia observed from week 8. Clinically, pain peaked at 8/10 on the Visual Analogue Scale (VAS) at week 32 and declined to 3/10 three months after explantation. The parallel reduction of oedema, restoration of vascular flow and normalisation of cytokine profiles accompanied pain relief, supporting a unified neurovascular inflammatory circuit consistent with the MNOH model [24-36].
Interpretation of Results. The presented case demonstrates that hypersensitivity to metallic implants can significantly impair bone healing even in the absence of infection. Prolonged local inflammation, wound leakage, swelling, and persistent pain reflected an aseptic immune reaction to metal corrosion products, activating T-cell inflammation, suppressing osteogenesis, and delaying callus formation. Similar mechanisms were observed by Dapunt et al. (2014), who reported local expression of cathepsin K, CD14, and CD3, along with pro-inflammatory and osteoclast-inducing cytokines (CXCL8, IL-1β, CXCL2, MRP-14), creating a potent osteolytic microenvironment; systemic CD4⁺ T-cell activation further supports T-cell involvement in metallosis progression [37]. Importantly, meticulous postoperative wound care – particularly when managed at home – is critical to prevent secondary complications and limit additional inflammatory triggers [38, 39].
An important diagnostic criterion was the absence of systemic and laboratory signs of infection while local inflammatory symptoms persisted. After implant removal, the rapid reduction of pain and inflammation and restoration of bone healing confirmed the immunological nature of the complication. Similar conclusions were reached by Wawrzynski J., Gil J.A., Goodman A.D., Waryasz G.R. (2017). Wawrzynski et al. note that surgical removal of the metal trigger leads to resolution of the hypersensitivity reaction [3]. The authors describe the case of a 12-year-old child with systemic allergic dermatitis caused by implantation of a nickel-cobalt plate, in whom the dermatitis rapidly resolved after implant removal [3]. They also report data on five patients with fibromyalgia and chronic fatigue syndrome, in whom all symptoms disappeared after removal of the metal implant. The researchers emphasize that in early clinical cases, eczematous dermatitis completely resolved within 72 hours after removal of stainless-steel implants [3]. Accordingly, returning to the results of our study, it can be concluded that the case highlights the need for early recognition of hypersensitivity as a potential cause of osteosynthesis failure and the importance of an individualized approach and careful postoperative monitoring.
The decision to remove the implant 8 months after osteosynthesis was based on a multicriteria assessment: (1) persistent pain (VAS 8/10) and chronic inflammatory signs without infection; (2) absence of radiological callus progression over 32 weeks despite stable fixation; (3) Th1/Th17 skewing, elevated serum Ni²⁺/Cr³⁺, and Ni-specific IgE. The 8-month timing balanced adequate mechanical stability with timely elimination of the immune trigger, in line with reports of increased refracture risk after early removal (<6 months) [32] and progressive RANKL/OPG imbalance with delayed extraction. Rapid normalization of cytokines and T-cell subpopulations after removal confirmed the appropriateness of the clinical decision in this patient.
CONCLUSIONS
1. In this case, chronic hypersensitivity to a 316L stainless steel implant was associated with persistent aseptic inflammation, Th1/Th17 polarisation (IFN-γ 8.2 pg/mL; IL-17A 6.3 pg/mL), RANKL/OPG imbalance, and absent radiological callus progression for 8 months, reflecting immune-mediated osteogenesis suppression.
2. The diagnostic profile included severe pain (VAS 8/10 at week 32), local hyperaemia and exudation without infection (negative cultures; normal WBC, CRP, ESR), Th1/Th17 skewing (Th17: 8.9%; Treg: 3.6%), elevated serum Ni²⁺ (8.6 µg/L) and Cr³⁺ (4.1 µg/L), and Ni-specific IgE class 2, consistent with a multicriteria DTH algorithm.
3. Preoperative allergological screening was not performed, but retrospective detection of Ni-specific IgE and early Th1/Th17 skewing suggests possible latent sensitisation prior to implantation.
4. Implant removal led to normalisation of cytokines (IFN-γ: 8.2→1.8 pg/mL; IL-17A: 6.3→1.1 pg/mL), restoration of Th1/Th17/Treg balance, improved microcirculation, and callus formation, confirming reversibility of metal-induced immune complications.
Contributors:
Bondarenko Ya.D. – conceptualization, methodology, writing – original draft, investigation, data curation, resources;
Stroiev M.Yu. – writing – review & editing, validation
Lytvynenko M.I. – supervision, writing – review & editing;
Bitchuk M.D. – writing – review & editing, formal analysis.
Funding. This research received no external funding.
Conflict of interests. The authors declare no conflict of interest.
REFERENCES
1. Shen M, Tejwani N. Open tibial shaft fracture fixation strategies: intramedullary nailing, external fixation, and plating. OTA Int. 2024;7(4 Suppl):e316. doi: https://doi.org/10.1097/OI9.0000000000000316
2. Rodrigues FL, Ferrari ALM, Faria FF, Pinto RLE, Lopes MF, Santos MEA, et al. Epidemiology and outcomes of intramedullary nailing for tibial diaphyseal fractures: a retrospective multicenter cohort study. Cureus. 2025;17(4):e82894. doi: https://doi.org/10.7759/cureus.82894
3. Wawrzynski J, Gil JA, Goodman AD, Waryasz GR. Hypersensitivity to orthopedic implants: a review of the literature. Rheumatol Ther. 2017;4(1):45-56. doi: https://doi.org/10.1007/s40744-017-0062-6
4. Kruckeberg BM, Ridley TJ, Reichel LM. Severe metal hypersensitivity following internal fixation of an ankle fracture: a case report. JBJS Case Connect. 2017;7(2):e38. doi: https://doi.org/10.2106/JBJS.CC.16.00202
5. Teo WZW, Schalock PC. Metal hypersensitivity reactions to orthopedic implants. Dermatol Ther (Heidelb). 2017;7(1):53-64.
doi: https://doi.org/10.1007/s13555-016-0162-1
6. Magone K, Luckenbill D, Goswami T. Metal ions as inflammatory initiators of osteolysis. Arch Orthop Trauma Surg. 2015;135(5):683-95. doi: https://doi.org/10.1007/s00402-015-2196-8
7. Sansone V, Pagani D, Melato M. The effects on bone cells of metal ions released from orthopaedic implants. A review. Clin Cases Miner Bone Metab. 2013;10(1):34-40. doi: https://doi.org/10.11138/ccmbm/2013.10.1.034
8. Furrer S, Scherer Hofmeier K, Grize L, Bircher AJ. Metal hypersensitivity in patients with orthopaedic implant complications: a retrospective clinical study. Contact Dermatitis. 2018;79(2):91-8. doi: https://doi.org/10.1111/cod.13032
9. Schultzel M, Klein CM, Demirjian M, Blout C, Itamura JM. Incidence of metal hypersensitivity in orthopedic surgical patients who self-report hypersensitivity history. Perm J. 2020;24:19.091. doi: https://doi.org/10.7812/TPP/19.091
10. Metal hypersensitivity in orthopaedic implants. Position statement. Canadian Orthopaedic Association [Internet]. 2025 [cited 2025 Oct 23]. Available from: https://coa-aco.org/wp-content/uploads/2025/11/PUBLIC-Metal-Hypersensitivity-EN.pdf
11. Practical guidance for the evaluation and management of metal implant allergy. Clinical guidance document. CeramTec Medical [Internet]. 2025 [cited 2025 Oct 23]. Available from: https://www.ceramtec-group.com
12. Thomas P, Arenberger P., Bader R., et al. A literature review and expert consensus statement on management of metal implant allergy. J Eur Acad Dermatol Venereol. 2024;38(6):1123-35. doi: https://doi.org/10.1111/jdv.20026
13. Moldovan F. Sterile inflammatory response and surgery-related trauma in elderly patients with subtrochanteric fractures. Biomedicines. 2024;12(2):354. doi: https://doi.org/10.3390/biomedicines12020354
14. Mervis JS. The impact of chronic wound exudate on the patient, clinician and payer: addressing the challenges with foam dressings. Int Wound J. 2025;22 (Suppl 1):e70369. doi: https://doi.org/10.1111/iwj.70369
15. R AS, Nambi N, Radhakrishnan L, Prasad MK, Ramkumar KM. Neutrophil migration is a crucial factor in wound healing and the pathogenesis of diabetic foot ulcers: insights into pharmacological interventions. Curr Vasc Pharmacol. 2024 Oct 30. Epub ahead of print. doi: https://doi.org/10.2174/0115701611308960241014155413
16. Rizo-Téllez SA, Filep JG. Beyond host defense and tissue injury: the emerging role of neutrophils in tissue repair. Am J Physiol Cell Physiol. 2024;326(3):C661-83. doi: https://doi.org/10.1152/ajpcell.00652.2023
17. Rezaei AR, Zienkiewicz D, Rezaei AR. Surgical site infections: a comprehensive review. J Trauma Inj. 2025;38(2):71-81. doi: https://doi.org/10.20408/jti.2025.0019
18. Chang F, Yan L, Zha Y, Hong X, Zhu K, Fei Y, et al. Development of a wound epithelialization healing model: reducing the impact of contraction healing on the wound surface. J Burn Care Res. 2024;45(4):1016-25. doi: https://doi.org/10.1093/jbcr/irae065
19. Uberoi A, McCready-Vangi A, Grice EA. The wound microbiota: microbial mechanisms of impaired wound healing and infection. Nat Rev Microbiol. 2024;22(8):507-21. doi: https://doi.org/10.1038/s41579-024-01035-z
20. Dong J, Wang W, Zhou W, Zhang S, Li M, Li N, et al. Immunomodulatory biomaterials for implant-associated infections: from conventional to advanced therapeutic strategies. Biomater Res. 2022;26(1):72. doi: https://doi.org/10.1186/s40824-022-00326-x
21. Liu Y, Gao M, Yan Y, Wang X, Dong Z, Cheng L, et al. Immunology in osseointegration after implantation. J Biomed Mater Res B Appl Biomater. 2025;113(4):e35566. doi: https://doi.org/10.1002/jbm.b.35566
22. Alhayaza R, Haque E, Karbasiafshar C, Sellke FW, Abid MR. The relationship between reactive oxygen species and endothelial cell metabolism. Front Chem. 2020;8:592688. doi: https://doi.org/10.3389/fchem.2020.592688
23. Thomas SR, Witting PK, Drummond GR. Redox control of endothelial function and dysfunction: molecular mechanisms and therapeutic opportunities. Antioxid Redox Signal. 2008;10(10):1713-65. doi: https://doi.org/10.1089/ars.2008.2027
24. Chen W, Zhu WQ, Qiu J. Impact of exogenous metal ions on peri-implant bone metabolism: a review. RSC Adv. 2021;11(22):13152-63. doi: https://doi.org/10.1039/d0ra09395e
25. Marek-Jozefowicz L, Nedoszytko B, Grochocka M, Żmijewski MA, Czajkowski R, Cubała WJ, et al. Molecular mechanisms of neurogenic inflammation of the skin. Int J Mol Sci. 2023;24(5):5001. doi: https://doi.org/10.3390/ijms24055001
26. Veruva SY, Lanman TH, Isaza JE, Freeman TA, Kurtz SM, Steinbeck MJ. Periprosthetic UHMWPE wear debris induces inflammation, vascularization, and innervation after total disc replacement in the lumbar spine. Clin Orthop Relat Res. 2017;475(5):1369-81. doi: https://doi.org/10.1007/s11999-016-4996-8
27. Mashaghi A, Marmalidou A, Tehrani M, Grace PM, Pothoulakis C, Dana R. Neuropeptide substance P and the immune response. Cell Mol Life Sci. 2016;73(22):4249-64. doi: https://doi.org/10.1007/s00018-016-2293-z
28. Lampa J, Westman M, Kadetoff D, Agréus AN, Le Maître E, Gillis-Haegerstrand C, et al. Peripheral inflammatory disease associated with centrally activated IL-1 system in humans and mice. Proc Natl Acad Sci USA. 2012;109(31):12728-33. doi: https://doi.org/10.1073/pnas.1118748109
29. Bondarenko YD, Kauk OI, Stetsenko SO, Rykhlik SV. Changes in the neuro-glial-vascular interface in metabolic intoxications in children (based on acetonemic syndrome and hyperammonemia). Ukr Neurosurg J. 2025;31(4):3-10. doi: https://doi.org/10.25305/unj.331349
30. Bondarenko Y, Kauk O, Stetsenko S, Pliten O. Neuro-glio-capillary dysfunction in children with respiratory infections: early clinical markers and the role of outpatient screening. Psychiatry Neurol Med Psychol. 2025;12(4(30)):449-71. doi: https://doi.org/10.26565/2312-5675-2025-30-03
31. Ponce-Lopez T. Peripheral inflammation and insulin resistance: their impact on blood-brain barrier integrity and glia activation in Alzheimer's disease. Int J Mol Sci. 2025;26(9):4209. doi: https://doi.org/10.3390/ijms26094209
32. Yao CK, Lin KC, Tarng YW, Chang WN, Renn JH. Removal of forearm plate leads to a high risk of refracture: decision regarding implant removal after fixation of the forearm and analysis of risk factors of refracture. Arch Orthop Trauma Surg. 2014;134(12):1691-7. doi: https://doi.org/10.1007/s00402-014-2079-4
33. Schilrreff P, Alexiev U. Chronic inflammation in non-healing skin wounds and promising natural bioactive compounds treatment. Int J Mol Sci. 2022;23(9):4928. doi: https://doi.org/10.3390/ijms23094928
34. Hallab NJ, Jacobs JJ. Biologic effects of implant debris. Bull NYU Hosp Jt Dis. 2009;67(2):182-8. PMID: 19583551.
35. Samelko L, Landgraeber S, McAllister K, Jacobs J, Hallab NJ. Cobalt alloy implant debris induces inflammation and bone loss primarily through danger signaling, not TLR4 activation: implications for DAMP-ening implant related inflammation. PloS One. 2016;11(7):e0160141. doi: https://doi.org/10.1371/journal.pone.0160141
36. He H, Qiao Y, Zhou Q, Wang Z, Chen X, Liu D, et al. Iron overload damages the endothelial mitochondria via the ROS/ADMA/DDAHII/eNOS/NO pathway. Oxid Med Cell Longev. 2019;2019:2340392. doi: https://doi.org/10.1155/2019/2340392
37. Dapunt U, Giese T, Lasitschka F, Reinders J, Lehner B, Kretzer JP, et al. On the inflammatory response in metal-on-metal implants. J Transl Med. 2014;12:74. doi: https://doi.org/10.1186/1479-5876-12-74
38. Bondarenko YD, Kotovshchykov MS, Dotsenko VV, Kulyk DYe. Choice between modern wound dressings in pediatric care: Hydrogel, hydrocolloids, silver-containing dressings. The Ukrainian Journal of Clinical Surgery. 2025;92(6):91-8. doi: https://doi.org/10.26779/2786-832X.2025.6.91
39. Bondarenko YD, Kulyk DYe, Kotovshchykov MS, Dotsenko VV. Treatment of wounds in children in outpatient settings without complete sterility: Tactical decisions and safe algorithms. The Ukrainian Journal of Clinical Surgery. 2026;93(1):79-89. doi: https://doi.org/10.26779/2786-832X.2026.1.79


 UK
UK  EN
EN